What points to design, simplicity, or complexity?
https://reasonandscience.catsboard.com/t3250-what-points-to-design-simplicity-or-complexity
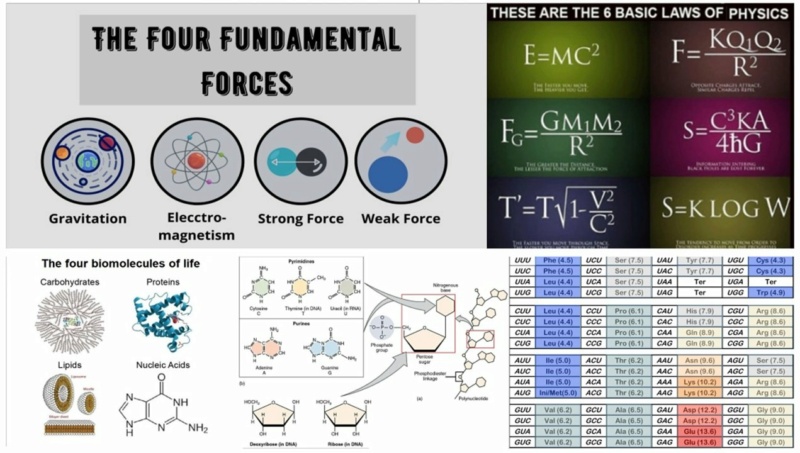
I hear from time to time, that simplicity, not complexity, is the hallmark of design. I think that is what we can observe in the universe. All the awe-inspiring diversity of stars, galaxies, planets, life forms, ecology, landscapes, and so forth is built based on just four fundamental forces of energy, six fundamental mathematical equations, four Building blocks of life, a genetic alphabet of four letters, and a genetic code, that assigns 64 códon words to 20 amino acids. So it is basically both, designed simplicity, combined with the complexity that comes from it, that points to design.
On top of that, physical forces and laws are fine-tuned. And the genetic code and the amino acid set are optimal amongst millions of less adequate alternatives.
The fundamental forces must be finely adjusted to the extreme to permit life
https://reasonandscience.catsboard.com/t1339-fine-tuning...
The laws of physics must also be finely adjusted to permit carbon-based life
https://reasonandscience.catsboard.com/t1336-laws-of...
No DNA double Helix, no life
And that depends on the hydrogen bond forces for Watson-Crick base pairing which must also be just right, and they depend on the isomeric configuration of the nucleobases of purines and pyrimidines. There is almost an infinite number of possible setups.
https://reasonandscience.catsboard.com/t2591-biochemical...
If we employ weightings to allow for biases in translation, then only 1 in every million random alternative genetic codes generated is more efficient than the natural code.
https://pubmed.ncbi.nlm.nih.gov/9732450/
In 2015, a scientific investigation was performed to test 100 million different sets of twenty amino acids. They measured three chemical properties: size, charge, and hydrophobicity, and compared it to the standard amino acid alphabet. Only six are better able to explore “chemistry space” than the twenty amino acids that life uses. That suggests that life’s set of amino acids is finely tuned to one part in 16 million.
https://www.nature.com/articles/srep09414
Parsimony, Fine-tuning, and optimality of sets =
https://reasonandscience.catsboard.com/t3250-what-points-to-design-simplicity-or-complexity
I hear from time to time, that simplicity, not complexity, is the hallmark of design. I think that is what we can observe in the universe. All the awe-inspiring diversity of stars, galaxies, planets, life forms, ecology, landscapes, and so forth is built based on just four fundamental forces of energy, six fundamental mathematical equations, four Building blocks of life, a genetic alphabet of four letters, and a genetic code, that assigns 64 códon words to 20 amino acids. So it is basically both, designed simplicity, combined with the complexity that comes from it, that points to design.
On top of that, physical forces and laws are fine-tuned. And the genetic code and the amino acid set are optimal amongst millions of less adequate alternatives.
The fundamental forces must be finely adjusted to the extreme to permit life
https://reasonandscience.catsboard.com/t1339-fine-tuning...
The laws of physics must also be finely adjusted to permit carbon-based life
https://reasonandscience.catsboard.com/t1336-laws-of...
No DNA double Helix, no life
And that depends on the hydrogen bond forces for Watson-Crick base pairing which must also be just right, and they depend on the isomeric configuration of the nucleobases of purines and pyrimidines. There is almost an infinite number of possible setups.
https://reasonandscience.catsboard.com/t2591-biochemical...
If we employ weightings to allow for biases in translation, then only 1 in every million random alternative genetic codes generated is more efficient than the natural code.
https://pubmed.ncbi.nlm.nih.gov/9732450/
In 2015, a scientific investigation was performed to test 100 million different sets of twenty amino acids. They measured three chemical properties: size, charge, and hydrophobicity, and compared it to the standard amino acid alphabet. Only six are better able to explore “chemistry space” than the twenty amino acids that life uses. That suggests that life’s set of amino acids is finely tuned to one part in 16 million.
https://www.nature.com/articles/srep09414
Parsimony, Fine-tuning, and optimality of sets =




