The Atom symbol - used by who has good reasons to use it
Stephen C. Meyer: The return of the God hypothesis, page 185
For instance, to make life possible, the masses of the fundamental particles must meet an exacting combination of constraints. In the previous chapter, I discussed the fine-tuning of the masses of the two naturally occurring quarks, the up quark and down quark, in relation to the range of expected possible values. Recall that the fine-tuning of the masses of those quarks is considerable—1 part in 10^21 . In addition, the difference in masses between the quarks cannot exceed one megaelectron volt, the equivalent of one-thousandth of 1 percent of the mass of the largest known quark, without producing either a neutron-only or a proton-only universe, both exceedingly boring and incompatible with life and even with simple chemistry. Equally problematic, increasing the mass of electrons by a factor of 2.5 would result in all the protons in all the atoms capturing all the orbiting electrons and turning them into neutrons. In that case, neither atoms, nor chemistry, nor life could exist. What’s more, the mass of the electron has to be less than the difference between the masses of the neutron and the proton and that difference represents fine-tuning of roughly 1 part in a 1000. In addition, if the mass of a special particle known as a neutrino were increased by a factor of 10, stars and galaxies would never have formed. The mass of a neutrino is about one-millionth that of an electron, so the allowable change is minuscule compared to its possible range. The combination of all these precisely fine-tuned conditions—including the fine-tuning of the laws and constants of physics, the initial arrangement of matter and energy, and various other contingent features of the universe —presents a remarkably restrictive set of criteria. These requirements for the existence of life, again defying our ability to describe their extreme improbability, have seemed to many physicists to require some explanation.
Strikingly, the masses of “up quarks” and “down quarks,” the constituent parts of protons and neutrons, must have precise values to allow for the production of the elements, including carbon, essential for a life-friendly universe. Indeed, the masses of these quarks must have simultaneously nine different conditions for the right nuclear reactions to have occurred in the early universe. The “right” reactions are ones that would produce the right elements (such as carbon and oxygen) in the right abundance necessary for life. The fine-tuning of the masses of these two naturally occurring quarks in relation to the range of expected possible values for the mass of any fundamental particle is exquisite. Physicists conceive of that range as extending between a mass of zero and the so-called Planck mass, an important unit of measure in quantum physics. But the value of the “up quark” must have a precise mass of between zero and just one billion trillionth of the Planck mass, corresponding to a fine-tuning of roughly 1 part in 10^21. The mass of the “down quark” must have a similarly precise fine-tuning.
Luke A. Barnes The Fine-Tuning of the Universe for Intelligent Life June 11, 2012
https://arxiv.org/pdf/1112.4647.pdf
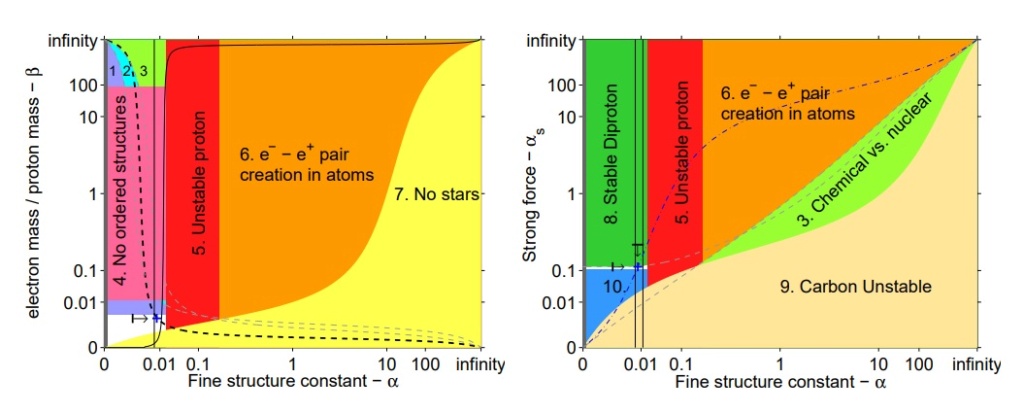
The life-permitting region (shown in white) in the (α, β) (left) and (α, αs) (right) parameter space, with other constants held at their values in our universe. Our universe is shown as a blue cross.
1. For hydrogen to exist — to power stars and form water and organic compounds — we must have me < mn − mp. Otherwise, the electron will be captured by the proton to form a neutron
2. For stable atoms, we need the radius of the electron orbit to be significantly larger than the nuclear radius.
3. We require that the typical energy of chemical reactions is much smaller than the typical energy of nuclear reactions. This ensures that the atomic constituents of chemical species maintain their identity in chemical reactions.
4. Unless β 1/4 << 1, stable ordered molecular structures (like chromosomes) are not stable. The atoms will too easily stray from their place in the lattice and the substance will spontaneously melt
5. The stability of the proton requires α . (md − mu)/141 MeV, so that the extra electromagnetic mass-energy of a proton relative to a neutron is more than counter-balanced by the bare quark masses
6. Unless α << 1, the electrons in atoms and molecules are unstable to pair creation. The limit shown is α < 0.2..
7. As in Equation 10, stars will not be stable unless β & α 2/100.
8. Unless αs/αs,0 . 1.003 + 0.031α/α0, the diproton has a bound state, which affects stellar burning and big bang nucleosynthesis.
9. Unless αs . 0.3α 1/2 , carbon and all larger elements are unstable
10. Unless αs/αs,0 & 0.91, the deuteron is unstable and the main nuclear reaction in stars (pp) does not proceed. A similar effect would be achieved35 unless md − mu + me < 3.4 MeV which makes the pp reaction energetically unfavourable. This region is numerically very similar to Region 1 in the left plot.
Atheists and proponents of philosophical naturalism use the Atom symbol with no justification - the Atom bears amazing evidence of design - the atomic forces must be finely tuned, otherwise, our universe would host no stars, no planets, no life.
For example, fine-tuning:
Ratio of Electrons: Protons 1:10^37
Ratio of Electromagnetic Force: Gravity 1:10^40
The electron, the proton, and the quark are all entities within the realm of particle hence quantum physics. All three carry an electrical charge. All three have mass. After those observations, things get interesting, or messy, depending on your point of view. The electric charge of the proton is exactly equal and the opposite of the electric charge on the electron, despite the proton being nearly 2000 times more massive. There’s no set-in-concrete theoretical reason why this should be so. It cannot be determined from first principles, only experimentally measured. 1
An electron has a negative charge exactly equal and opposite to that of a proton. Note: the charge is exactly equal, even though the proton has a far greater mass than the electron (some 2000 times heavier in fact, not that there has to be of necessity any relationship between mass and charge).
Now that’s strange since the electron is a fundamental particle but the positively charged proton is a composite particle, made up of a trio of quarks (as is the neutron with no net charge). The proton has two quarks each with a positive 2/3rds charge (up quark) and one quark with a negative 1/3rd charge (down quark) for an overall balance of one positive charge. (The neutron, on the other hand, has one up quark with a positive 2/3rds charge and two down quarks each with a negative 1/3rd charge, for an overall balance of zero charge – neither positive nor negative.)
Now you might suggest that an electron might be a fusion of a trio of down quarks, each with a negative 1/3rd charge, except the electron, again, isn’t a composite particle, and the mass is all wrong for that scenario. If an electron were a composite of a trio of down quarks, each with a minus 1/3rd charge, the electron would be thirty times more massive than it is – not something particle physicists would fail to take notice of.
Further, the force particle that governs the electron is the photon; that which governs the quarks inside the proton and the neutron is the gluon, which further differentiates the two things – quarks and electrons. In any event, if you could have a composite particle of a trio of negative 1/3rd down quarks, if that were the case, and it is the case, and it’s called the Negative Delta, you’d also need a composite particle that’s the fusion of a trio of positive 2/3rds up quarks for an overall charge of plus two. To the best of my knowledge, there is only one such critter in the particle zoo and it’s called the Doubly Positive Delta. I’m sure you’ve never heard of these Delta particles, which goes to show how much bearing or impact they have on life, the Universe, and everything.
In case you were wondering, there would be an anti-quark of minus 2/3rds charge, and an anti-quark of a positive 1/3rd charge, to yield an antiproton and an anti-neutron. The anti-proton would of course have an equal and opposite charge to the anti-electron (which has a formal name – the position). So things are equally as mysterious in the realm of the anti-world.
Question: How do you get 1/3rd or 2/3rds of an electric charge in any event? Of course one could just multiply by three and that does away with the fractions, but that doesn’t resolve the larger issues, like for that matter, what exactly is electric charge and how does it come to be?
Gravity:
How finely tuned is gravity? -- Well, the strength of gravity could be at any one of 14 billion billion billion settings, but there is only one setting which is adequate (and optimal) for a universe with intelligent life to exist.
-- To illustrate: This is as if you had a measuring tape with one-inch sections stretched across the known universe, it would be 14 billion billion billion inches long, and only one or two of those inches in the middle is the optimal strength setting for gravity. If you moved the strength setting to the right or left just a couple of inches, then intelligent life could not exist (though bacterial life might survive with gravity stronger or weaker by one setting up or down).
THE PROBABILITY: Although the force of gravity could obviously have attained a large number of wrong magnitude-ranges, the chance of it being correct for intelligent life to exist, is one chance out of 14 billion billion billion. --Thus, we can conservatively say that it was about one chance out of 1,000,000,000,000,000,000,000 (or 1 out of 10^21, or 1 out of a billion trillion) that the force of gravity might have randomly attained such an advantageous strength for the making of life-necessary elements in the stars.
Imagine stretching a measuring tape across the entire known universe. Now imagine one particular mark on the tape represents the correct degree of gravitational force required to create the universe we have. If this mark were moved more than an inch from its location (on a measuring tape spanning the entire universe), the altered gravitational force would prevent our universe from coming into existence
The Strong Nuclear Force
If the strong nuclear force were very slightly weaker by just one part in 10,000 billion billion billion billion, then protons and neutrons would not stick together, and the only element possible in the universe, would be hydrogen only. There would be no stars and no planets or life in the universe. (Ref., Dr. Robin Collins of Messiah College).
However, if the strong nuclear force were slightly too strong by the same fraction amount, the protons and neutrons would tend to stick together so much that there would basically only be heavy elements, but no hydrogen at all --If this were the case, then life would also not be possible, because hydrogen is a key element in water and in all life-chemistry.
THE PROBABILITY: If the strong nuclear force were slightly weaker or stronger than it in fact is, then life would be impossible. Therefore, we can very conservatively say that it was about one chance out of 1,000,000,000,000 (1 out of a trillion) that the strong nuclear force might have randomly possessed the correct strength to make life possible in our cosmos.
The Weak Nuclear Force
Our hydrogen-burning Sun consumes its nuclear fuel more than one hundred times more slowly than a pure helium star of comparable mass. A helium star of an appropriate mass wouldn’t last nearly long enough for life to develop. Not that life would ever develop around such a star anyway: it would contain no water or organic compounds, making the formation of life on any timescale impossible. It’s not even clear that stars could form from contracting clouds of gas in a universe of pure helium. Unlike hydrogen, helium does not form molecules, which are the primary means by which dense interstellar clouds cool, and thereby contract to form stars. 2
The weak nuclear force is what controls the rates at which radioactive elements decay. If this force were slightly stronger, the matter would decay into the heavy elements in a relatively short time. However, if it were significantly weaker, all matter would almost totally exist in the form of the lightest elements, especially hydrogen and helium ---there would be (for example) virtually no oxygen, carbon or nitrogen, which are essential for life.
In addition, although heavier elements necessary for life are formed inside giant stars, those elements can only escape the cores of those stars when they explode in supernova explosions, however, such supernova explosions can only occur because the weak nuclear force is exactly the right value. As Professor of astronomy, Paul Davies, describes this situation: "If the weak interaction were slightly weaker, the neutrinos would not be able to exert enough pressure on the outer envelope of the star to cause the supernova explosion. On the other hand, if it were slightly stronger, the neutrinos would be trapped inside the core, and rendered impotent" (My emphasis.) (ref. P.C.W. Davies, The Accidental Universe, London, 1982, p.68.)
THE PROBABILITY: Considering the fine-tuning of the weak nuclear force for both the rate of radioactive decay as well as the precise value required to allow supernova explosions, it is probably conservative to say that it was one chance out of 1000 that the weak nuclear force was at the right strength to permit these processes so that life would be possible.
The Electromagnetic Force
Electrons are bound by the electromagnetic force to atomic nuclei, and their orbital shapes and their influence on nearby atoms with their electrons is described by quantum mechanics. The electromagnetic force governs the processes involved in chemistry, which arise from interactions between the electrons of neighboring atoms. 1
If the electromagnetic force (exerted by electrons) were somewhat stronger, electrons would adhere to atoms so tightly that atoms would not share their electrons with each other ---and the sharing of electrons between atoms is what makes chemical bonding possible so that atoms can combine into molecules (e.g., water) so that life can exist. However, if the electromagnetic force were somewhat weaker, then atoms would not hang onto electrons enough to cause any bonding between atoms, and thus, compounds would never hold together. In addition, this fine-tuning of the electromagnetic force must be even more stringent if more and more elements are to be able to bond together into many different types of molecules.
THE PROBABILITY: Considering the range of electromagnetic force that might have occurred, it is reasonable to say that the probability of the electromagnetic force being balanced at the right level for many thousands of compounds to function for the making of chemical compounds necessary for life, is one chance out of 1000.
So, the atom symbol would be well appropriated to be used by proponents of intelligent design - and not atheists.
Fine-tuning of the fundamental forces of the universe
https://reasonandscience.catsboard.com/t1339-fine-tuning-of-the-fundamental-forces-of-the-universe
Quark fine-tuning
https://reasonandscience.catsboard.com/t2538-quark-fine-tuning
The Force of Gravity
https://reasonandscience.catsboard.com/t1366-the-force-of-gravity-evidence-of-fine-tuning
A Classic Formula For Pi Has Been Discovered Hidden in Hydrogen Atoms
https://reasonandscience.catsboard.com/t2554-a-classic-formula-for-pi-has-been-discovered-hidden-in-hydrogen-atoms
1. https://www.tapatalk.com/groups/vixra/electric-charge-an-example-of-fine-tuning-t121.html

Stephen C. Meyer: The return of the God hypothesis, page 185
For instance, to make life possible, the masses of the fundamental particles must meet an exacting combination of constraints. In the previous chapter, I discussed the fine-tuning of the masses of the two naturally occurring quarks, the up quark and down quark, in relation to the range of expected possible values. Recall that the fine-tuning of the masses of those quarks is considerable—1 part in 10^21 . In addition, the difference in masses between the quarks cannot exceed one megaelectron volt, the equivalent of one-thousandth of 1 percent of the mass of the largest known quark, without producing either a neutron-only or a proton-only universe, both exceedingly boring and incompatible with life and even with simple chemistry. Equally problematic, increasing the mass of electrons by a factor of 2.5 would result in all the protons in all the atoms capturing all the orbiting electrons and turning them into neutrons. In that case, neither atoms, nor chemistry, nor life could exist. What’s more, the mass of the electron has to be less than the difference between the masses of the neutron and the proton and that difference represents fine-tuning of roughly 1 part in a 1000. In addition, if the mass of a special particle known as a neutrino were increased by a factor of 10, stars and galaxies would never have formed. The mass of a neutrino is about one-millionth that of an electron, so the allowable change is minuscule compared to its possible range. The combination of all these precisely fine-tuned conditions—including the fine-tuning of the laws and constants of physics, the initial arrangement of matter and energy, and various other contingent features of the universe —presents a remarkably restrictive set of criteria. These requirements for the existence of life, again defying our ability to describe their extreme improbability, have seemed to many physicists to require some explanation.
Strikingly, the masses of “up quarks” and “down quarks,” the constituent parts of protons and neutrons, must have precise values to allow for the production of the elements, including carbon, essential for a life-friendly universe. Indeed, the masses of these quarks must have simultaneously nine different conditions for the right nuclear reactions to have occurred in the early universe. The “right” reactions are ones that would produce the right elements (such as carbon and oxygen) in the right abundance necessary for life. The fine-tuning of the masses of these two naturally occurring quarks in relation to the range of expected possible values for the mass of any fundamental particle is exquisite. Physicists conceive of that range as extending between a mass of zero and the so-called Planck mass, an important unit of measure in quantum physics. But the value of the “up quark” must have a precise mass of between zero and just one billion trillionth of the Planck mass, corresponding to a fine-tuning of roughly 1 part in 10^21. The mass of the “down quark” must have a similarly precise fine-tuning.
Luke A. Barnes The Fine-Tuning of the Universe for Intelligent Life June 11, 2012
https://arxiv.org/pdf/1112.4647.pdf

The life-permitting region (shown in white) in the (α, β) (left) and (α, αs) (right) parameter space, with other constants held at their values in our universe. Our universe is shown as a blue cross.
1. For hydrogen to exist — to power stars and form water and organic compounds — we must have me < mn − mp. Otherwise, the electron will be captured by the proton to form a neutron
2. For stable atoms, we need the radius of the electron orbit to be significantly larger than the nuclear radius.
3. We require that the typical energy of chemical reactions is much smaller than the typical energy of nuclear reactions. This ensures that the atomic constituents of chemical species maintain their identity in chemical reactions.
4. Unless β 1/4 << 1, stable ordered molecular structures (like chromosomes) are not stable. The atoms will too easily stray from their place in the lattice and the substance will spontaneously melt
5. The stability of the proton requires α . (md − mu)/141 MeV, so that the extra electromagnetic mass-energy of a proton relative to a neutron is more than counter-balanced by the bare quark masses
6. Unless α << 1, the electrons in atoms and molecules are unstable to pair creation. The limit shown is α < 0.2..
7. As in Equation 10, stars will not be stable unless β & α 2/100.
8. Unless αs/αs,0 . 1.003 + 0.031α/α0, the diproton has a bound state, which affects stellar burning and big bang nucleosynthesis.
9. Unless αs . 0.3α 1/2 , carbon and all larger elements are unstable
10. Unless αs/αs,0 & 0.91, the deuteron is unstable and the main nuclear reaction in stars (pp) does not proceed. A similar effect would be achieved35 unless md − mu + me < 3.4 MeV which makes the pp reaction energetically unfavourable. This region is numerically very similar to Region 1 in the left plot.
Atheists and proponents of philosophical naturalism use the Atom symbol with no justification - the Atom bears amazing evidence of design - the atomic forces must be finely tuned, otherwise, our universe would host no stars, no planets, no life.
For example, fine-tuning:
Ratio of Electrons: Protons 1:10^37
Ratio of Electromagnetic Force: Gravity 1:10^40
The electron, the proton, and the quark are all entities within the realm of particle hence quantum physics. All three carry an electrical charge. All three have mass. After those observations, things get interesting, or messy, depending on your point of view. The electric charge of the proton is exactly equal and the opposite of the electric charge on the electron, despite the proton being nearly 2000 times more massive. There’s no set-in-concrete theoretical reason why this should be so. It cannot be determined from first principles, only experimentally measured. 1
An electron has a negative charge exactly equal and opposite to that of a proton. Note: the charge is exactly equal, even though the proton has a far greater mass than the electron (some 2000 times heavier in fact, not that there has to be of necessity any relationship between mass and charge).
Now that’s strange since the electron is a fundamental particle but the positively charged proton is a composite particle, made up of a trio of quarks (as is the neutron with no net charge). The proton has two quarks each with a positive 2/3rds charge (up quark) and one quark with a negative 1/3rd charge (down quark) for an overall balance of one positive charge. (The neutron, on the other hand, has one up quark with a positive 2/3rds charge and two down quarks each with a negative 1/3rd charge, for an overall balance of zero charge – neither positive nor negative.)
Now you might suggest that an electron might be a fusion of a trio of down quarks, each with a negative 1/3rd charge, except the electron, again, isn’t a composite particle, and the mass is all wrong for that scenario. If an electron were a composite of a trio of down quarks, each with a minus 1/3rd charge, the electron would be thirty times more massive than it is – not something particle physicists would fail to take notice of.
Further, the force particle that governs the electron is the photon; that which governs the quarks inside the proton and the neutron is the gluon, which further differentiates the two things – quarks and electrons. In any event, if you could have a composite particle of a trio of negative 1/3rd down quarks, if that were the case, and it is the case, and it’s called the Negative Delta, you’d also need a composite particle that’s the fusion of a trio of positive 2/3rds up quarks for an overall charge of plus two. To the best of my knowledge, there is only one such critter in the particle zoo and it’s called the Doubly Positive Delta. I’m sure you’ve never heard of these Delta particles, which goes to show how much bearing or impact they have on life, the Universe, and everything.
In case you were wondering, there would be an anti-quark of minus 2/3rds charge, and an anti-quark of a positive 1/3rd charge, to yield an antiproton and an anti-neutron. The anti-proton would of course have an equal and opposite charge to the anti-electron (which has a formal name – the position). So things are equally as mysterious in the realm of the anti-world.
Question: How do you get 1/3rd or 2/3rds of an electric charge in any event? Of course one could just multiply by three and that does away with the fractions, but that doesn’t resolve the larger issues, like for that matter, what exactly is electric charge and how does it come to be?
Gravity:
How finely tuned is gravity? -- Well, the strength of gravity could be at any one of 14 billion billion billion settings, but there is only one setting which is adequate (and optimal) for a universe with intelligent life to exist.
-- To illustrate: This is as if you had a measuring tape with one-inch sections stretched across the known universe, it would be 14 billion billion billion inches long, and only one or two of those inches in the middle is the optimal strength setting for gravity. If you moved the strength setting to the right or left just a couple of inches, then intelligent life could not exist (though bacterial life might survive with gravity stronger or weaker by one setting up or down).
THE PROBABILITY: Although the force of gravity could obviously have attained a large number of wrong magnitude-ranges, the chance of it being correct for intelligent life to exist, is one chance out of 14 billion billion billion. --Thus, we can conservatively say that it was about one chance out of 1,000,000,000,000,000,000,000 (or 1 out of 10^21, or 1 out of a billion trillion) that the force of gravity might have randomly attained such an advantageous strength for the making of life-necessary elements in the stars.
Imagine stretching a measuring tape across the entire known universe. Now imagine one particular mark on the tape represents the correct degree of gravitational force required to create the universe we have. If this mark were moved more than an inch from its location (on a measuring tape spanning the entire universe), the altered gravitational force would prevent our universe from coming into existence
The Strong Nuclear Force
If the strong nuclear force were very slightly weaker by just one part in 10,000 billion billion billion billion, then protons and neutrons would not stick together, and the only element possible in the universe, would be hydrogen only. There would be no stars and no planets or life in the universe. (Ref., Dr. Robin Collins of Messiah College).
However, if the strong nuclear force were slightly too strong by the same fraction amount, the protons and neutrons would tend to stick together so much that there would basically only be heavy elements, but no hydrogen at all --If this were the case, then life would also not be possible, because hydrogen is a key element in water and in all life-chemistry.
THE PROBABILITY: If the strong nuclear force were slightly weaker or stronger than it in fact is, then life would be impossible. Therefore, we can very conservatively say that it was about one chance out of 1,000,000,000,000 (1 out of a trillion) that the strong nuclear force might have randomly possessed the correct strength to make life possible in our cosmos.
The Weak Nuclear Force
Our hydrogen-burning Sun consumes its nuclear fuel more than one hundred times more slowly than a pure helium star of comparable mass. A helium star of an appropriate mass wouldn’t last nearly long enough for life to develop. Not that life would ever develop around such a star anyway: it would contain no water or organic compounds, making the formation of life on any timescale impossible. It’s not even clear that stars could form from contracting clouds of gas in a universe of pure helium. Unlike hydrogen, helium does not form molecules, which are the primary means by which dense interstellar clouds cool, and thereby contract to form stars. 2
The weak nuclear force is what controls the rates at which radioactive elements decay. If this force were slightly stronger, the matter would decay into the heavy elements in a relatively short time. However, if it were significantly weaker, all matter would almost totally exist in the form of the lightest elements, especially hydrogen and helium ---there would be (for example) virtually no oxygen, carbon or nitrogen, which are essential for life.
In addition, although heavier elements necessary for life are formed inside giant stars, those elements can only escape the cores of those stars when they explode in supernova explosions, however, such supernova explosions can only occur because the weak nuclear force is exactly the right value. As Professor of astronomy, Paul Davies, describes this situation: "If the weak interaction were slightly weaker, the neutrinos would not be able to exert enough pressure on the outer envelope of the star to cause the supernova explosion. On the other hand, if it were slightly stronger, the neutrinos would be trapped inside the core, and rendered impotent" (My emphasis.) (ref. P.C.W. Davies, The Accidental Universe, London, 1982, p.68.)
THE PROBABILITY: Considering the fine-tuning of the weak nuclear force for both the rate of radioactive decay as well as the precise value required to allow supernova explosions, it is probably conservative to say that it was one chance out of 1000 that the weak nuclear force was at the right strength to permit these processes so that life would be possible.
The Electromagnetic Force
Electrons are bound by the electromagnetic force to atomic nuclei, and their orbital shapes and their influence on nearby atoms with their electrons is described by quantum mechanics. The electromagnetic force governs the processes involved in chemistry, which arise from interactions between the electrons of neighboring atoms. 1
If the electromagnetic force (exerted by electrons) were somewhat stronger, electrons would adhere to atoms so tightly that atoms would not share their electrons with each other ---and the sharing of electrons between atoms is what makes chemical bonding possible so that atoms can combine into molecules (e.g., water) so that life can exist. However, if the electromagnetic force were somewhat weaker, then atoms would not hang onto electrons enough to cause any bonding between atoms, and thus, compounds would never hold together. In addition, this fine-tuning of the electromagnetic force must be even more stringent if more and more elements are to be able to bond together into many different types of molecules.
THE PROBABILITY: Considering the range of electromagnetic force that might have occurred, it is reasonable to say that the probability of the electromagnetic force being balanced at the right level for many thousands of compounds to function for the making of chemical compounds necessary for life, is one chance out of 1000.
So, the atom symbol would be well appropriated to be used by proponents of intelligent design - and not atheists.
Fine-tuning of the fundamental forces of the universe
https://reasonandscience.catsboard.com/t1339-fine-tuning-of-the-fundamental-forces-of-the-universe
Quark fine-tuning
https://reasonandscience.catsboard.com/t2538-quark-fine-tuning
The Force of Gravity
https://reasonandscience.catsboard.com/t1366-the-force-of-gravity-evidence-of-fine-tuning
A Classic Formula For Pi Has Been Discovered Hidden in Hydrogen Atoms
https://reasonandscience.catsboard.com/t2554-a-classic-formula-for-pi-has-been-discovered-hidden-in-hydrogen-atoms
1. https://www.tapatalk.com/groups/vixra/electric-charge-an-example-of-fine-tuning-t121.html

Last edited by Otangelo on Wed Jun 23, 2021 5:40 pm; edited 1 time in total


