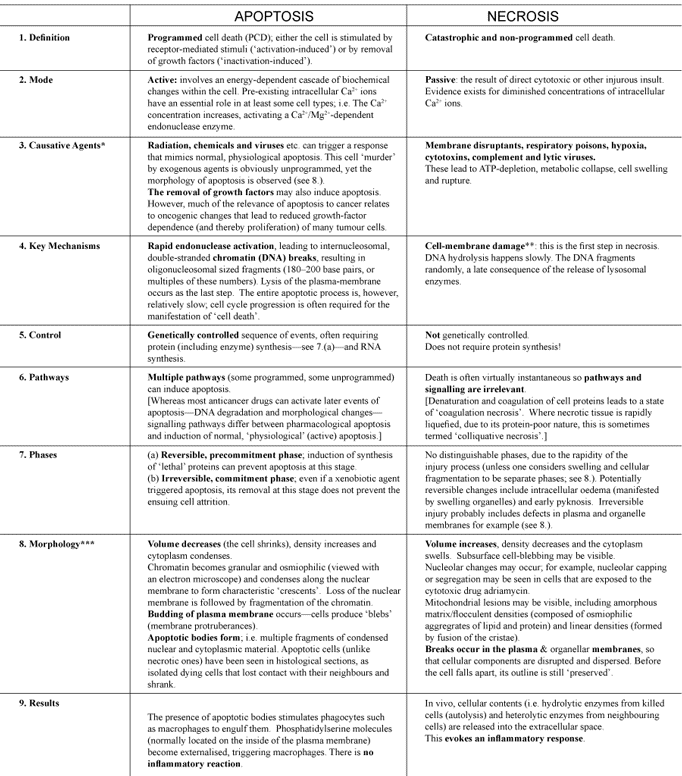
Apoptosis, Cell's essential mechanism of programmed suicide points to design
https://reasonandscience.catsboard.com/t2193-apoptosis-cell-s-essential-mechanism-of-programmed-suicide-points-to-design
Survival of the Fittest or Altruistic Suicide? 1
Like engineers carefully blowing up a bridge, cells have intricate, programmed suicide mechanisms. The signal is sent and an apparatus of destruction is activated. But suicide hardly fits the evolutionary narrative. Wasn’t this all about survival, reproductive advantages and leaving more offspring? Why would a cell evolve intricate and complex suicide machinery?
The answer is that suicide at the cellular level doesn’t kill the whole organism. Such self-destruction serves a range of purposes, from guiding development to keeping cancer at bay. In short, cell death in a multicellular organism can be a good thing.
But if cell suicide in multicellular organisms passes the evolutionary test, what about recent findings of suicide in unicellular organisms? New genome data from the Great Barrier Reef demosponge (Amphimedon queenslandica) reveals high levels of unexpected complexity, for this lowly sponge has an impressive complement of genes. As one proponent of evolution put it, “This flies in the face of what we think of early metazoan evolution.”
Another Darwinist asked perhaps an even more telling question. “What I want to know now,” he asked, “is what were all these genes doing prior to the advent of sponge?”
That’s a good question because some of those genes are for programmed suicide. What this sponge genome apparently tells us is that programmed cell death would have to have arisen in single-cell organisms. Suicide at the cellular level did kill the whole organism—and that doesn’t make evolutionary sense.
With evolution what we must believe is that programmed cell death did not arise in multicellular species, but in unicellular species. In other words, an intricate, highly complex, set of tools and signals somehow arose and, rather than leading to enhanced survival as evolution calls for, they led to destruction. Proponents of evolution will need yet another one of their just-so stories to rationalize this.
Programmed Cell Death
Cells have built-in error correction and repair mechanisms. Such mechanisms cannot be accounted for by any Darwinian hypothesis since error detection implies knowledge. Knowledge of correct system state and planning of appropriate repair measures.
Here’s one problem with the Darwinian “it just evolved” hypothesis :
Q: How can a repair mechanism arise without pre-knowledge of correct system state?
A: It cannot. Not when the damage caused interrupts and corrective facilities are this specifically complex
Implication? DNA had to be designed by an intelligent agent.
In software development, programmers build what we call “exception trapping” mechanisms.
Such mechanisms ‘watch’ a given function progress and trap errors (exceptional system events) when detected. The trapping code then directs program flow either to analysis functions and correctional code or if the error is minor simply continue processing after the code block that failed (caused the exception) and may possibly alert the user to a faulty input situation.
DNA has it’s own special codes for detecting and ‘catching’ the exceptions that occur. Triggered by a diverse range of cell signals.
Neo Darwinian evolution (NDE) cannot explain the existence of such built-in functions.
Worse (for NDE) : Not only is there repair of damage available to the cell’s system but there is even a last resort “correction” (but not repaired) measure called apoptosis – pre-programmed cell death!Apoptosis is “a type of cell death in which the cell uses specialized cellular machinery to kill itself; a cell suicide mechanism that enables metazoans to control cell number and eliminate cells that threaten the animal’s survival” (also plays a role in preventing cancer). Its a key process in multicellular organisms.
This too is not explicable under NDE.
So, in a more engineering-like term we can look at this process as something like a control feedback loop. Such ‘loops’ exist in many places in the cell; like the circadian oscillator (Paley's watch!) – “a clockwork mechanism that controls these global rhythms of transcription, chromosomal topology, and cell division.”
Well then, the evolutionary ancestor to that was what?
Darwinism utterly fails to predict sophisticated repair mechanisms like these. Only pre-knowledge of correct system state can foresee and then construct code sequences that operate repairs to or destruction of faulty parts in a complex machine.
DNA is such a machine.
Programmed cell death (PCD, or apoptosis) appears to be universally present and indeed should be regarded as one of the hallmarks of the multicellular state itself 2 From a purely systemic point of view, PCD in a multicellular organism appears to be just as inevitable as law enforcement in a state. In any differentiated community (of specialized cells or of citizens), rogue elements will necessarily emerge in as (pre)cancerous cells with impaired division control or as criminals with impaired social responsibility, and to protect the community, these need to be subdued or destroyed by dedicated agencies. Those agencies also contribute to the defense against invaders, such as viruses, pathogenic bacteria or rival states, and, at least in the case of PCD, to the normal development of the multicellular organism. Eukaryotes had to come up with a PCD systems in order for any complex form of multicellularity to emerge and, accordingly, for us to be here and ponder the mysteries of biological evolution, the origin of apoptosis among them.Thus, the question that needs to be addressed regarding the origin of eukaryotic PCD is not so much why, but how, i.e. where have the suitable components for the making of the apoptosis molecular machinery come from and what particular innovation(s) did it take to piece them together?
Koonin mentions a few possibilities, like co-option fro prokaryotes, horizontal gene transfer, or just evolution. None is convincing. So basically, no apoptosis, no multicellular life. That is a clear indication that an evolutionary, stepwise or evolutionary manner is not possible, but the mechanism had to be fully working from day 1.
The apoptosome: signaling platform of cell death
Recent work on the initial switches that trigger cell death has revealed surprising inventions of nature that ensure the ordered suicide of a cell that has been selected for demise. Particularly intriguing is how a signal — the release of cytochrome c from the mitochondria — is translated into the activation of the death cascade, which leads to a point of no return. Now there is new understanding of how this crucial process is delicately handled by a cytosolic signaling platform known as the apoptosome. The formation of the apoptosome and the activation of its effector, caspase-9, reveals a sophisticated mechanism that might be more common than was initially thought. 1
Cell-signaling pathways convert a stimulus into cellular action, and they generally have an initiation phase and an effector phase. The initiation phase usually transmits information from a ligand, through interaction with its receptor, to the first biochemical event, resulting in propagation of the pathway. The effector phase executes the biochemical events that result in the desired (often complex) cellular response. In contrast to signaling pathways that use phosphorylation, glycosylation, ubiquitylation or acetylation to alter protein properties, those that require proteolysis are irreversible, because cells are not known to possess a mechanism to re-ligate cut peptide bonds.
Apoptosis is such a pathway; indeed, one can appreciate that once a cell has made the decision to commit suicide, there is not much point in going back. Apoptosis pathways in mammals can have one of two initiation phases, and which pathway is selected depends on the nature of the death signal to be integrated
1. http://darwins-god.blogspot.com.br/search?q=multicellular
2. http://www.nature.com/cdd/journal/v9/n4/full/4400991a.html
https://reasonandscience.catsboard.com/t2193-apoptosis-cell-s-essential-mechanism-of-programmed-suicide-points-to-design
Survival of the Fittest or Altruistic Suicide? 1
Like engineers carefully blowing up a bridge, cells have intricate, programmed suicide mechanisms. The signal is sent and an apparatus of destruction is activated. But suicide hardly fits the evolutionary narrative. Wasn’t this all about survival, reproductive advantages and leaving more offspring? Why would a cell evolve intricate and complex suicide machinery?
The answer is that suicide at the cellular level doesn’t kill the whole organism. Such self-destruction serves a range of purposes, from guiding development to keeping cancer at bay. In short, cell death in a multicellular organism can be a good thing.
But if cell suicide in multicellular organisms passes the evolutionary test, what about recent findings of suicide in unicellular organisms? New genome data from the Great Barrier Reef demosponge (Amphimedon queenslandica) reveals high levels of unexpected complexity, for this lowly sponge has an impressive complement of genes. As one proponent of evolution put it, “This flies in the face of what we think of early metazoan evolution.”
Another Darwinist asked perhaps an even more telling question. “What I want to know now,” he asked, “is what were all these genes doing prior to the advent of sponge?”
That’s a good question because some of those genes are for programmed suicide. What this sponge genome apparently tells us is that programmed cell death would have to have arisen in single-cell organisms. Suicide at the cellular level did kill the whole organism—and that doesn’t make evolutionary sense.
With evolution what we must believe is that programmed cell death did not arise in multicellular species, but in unicellular species. In other words, an intricate, highly complex, set of tools and signals somehow arose and, rather than leading to enhanced survival as evolution calls for, they led to destruction. Proponents of evolution will need yet another one of their just-so stories to rationalize this.
Programmed Cell Death
Cells have built-in error correction and repair mechanisms. Such mechanisms cannot be accounted for by any Darwinian hypothesis since error detection implies knowledge. Knowledge of correct system state and planning of appropriate repair measures.
Here’s one problem with the Darwinian “it just evolved” hypothesis :
Q: How can a repair mechanism arise without pre-knowledge of correct system state?
A: It cannot. Not when the damage caused interrupts and corrective facilities are this specifically complex
Implication? DNA had to be designed by an intelligent agent.
In software development, programmers build what we call “exception trapping” mechanisms.
Such mechanisms ‘watch’ a given function progress and trap errors (exceptional system events) when detected. The trapping code then directs program flow either to analysis functions and correctional code or if the error is minor simply continue processing after the code block that failed (caused the exception) and may possibly alert the user to a faulty input situation.
DNA has it’s own special codes for detecting and ‘catching’ the exceptions that occur. Triggered by a diverse range of cell signals.
Neo Darwinian evolution (NDE) cannot explain the existence of such built-in functions.
Worse (for NDE) : Not only is there repair of damage available to the cell’s system but there is even a last resort “correction” (but not repaired) measure called apoptosis – pre-programmed cell death!Apoptosis is “a type of cell death in which the cell uses specialized cellular machinery to kill itself; a cell suicide mechanism that enables metazoans to control cell number and eliminate cells that threaten the animal’s survival” (also plays a role in preventing cancer). Its a key process in multicellular organisms.
This too is not explicable under NDE.
So, in a more engineering-like term we can look at this process as something like a control feedback loop. Such ‘loops’ exist in many places in the cell; like the circadian oscillator (Paley's watch!) – “a clockwork mechanism that controls these global rhythms of transcription, chromosomal topology, and cell division.”
Well then, the evolutionary ancestor to that was what?
Darwinism utterly fails to predict sophisticated repair mechanisms like these. Only pre-knowledge of correct system state can foresee and then construct code sequences that operate repairs to or destruction of faulty parts in a complex machine.
DNA is such a machine.
Programmed cell death (PCD, or apoptosis) appears to be universally present and indeed should be regarded as one of the hallmarks of the multicellular state itself 2 From a purely systemic point of view, PCD in a multicellular organism appears to be just as inevitable as law enforcement in a state. In any differentiated community (of specialized cells or of citizens), rogue elements will necessarily emerge in as (pre)cancerous cells with impaired division control or as criminals with impaired social responsibility, and to protect the community, these need to be subdued or destroyed by dedicated agencies. Those agencies also contribute to the defense against invaders, such as viruses, pathogenic bacteria or rival states, and, at least in the case of PCD, to the normal development of the multicellular organism. Eukaryotes had to come up with a PCD systems in order for any complex form of multicellularity to emerge and, accordingly, for us to be here and ponder the mysteries of biological evolution, the origin of apoptosis among them.Thus, the question that needs to be addressed regarding the origin of eukaryotic PCD is not so much why, but how, i.e. where have the suitable components for the making of the apoptosis molecular machinery come from and what particular innovation(s) did it take to piece them together?
Koonin mentions a few possibilities, like co-option fro prokaryotes, horizontal gene transfer, or just evolution. None is convincing. So basically, no apoptosis, no multicellular life. That is a clear indication that an evolutionary, stepwise or evolutionary manner is not possible, but the mechanism had to be fully working from day 1.
The apoptosome: signaling platform of cell death
Recent work on the initial switches that trigger cell death has revealed surprising inventions of nature that ensure the ordered suicide of a cell that has been selected for demise. Particularly intriguing is how a signal — the release of cytochrome c from the mitochondria — is translated into the activation of the death cascade, which leads to a point of no return. Now there is new understanding of how this crucial process is delicately handled by a cytosolic signaling platform known as the apoptosome. The formation of the apoptosome and the activation of its effector, caspase-9, reveals a sophisticated mechanism that might be more common than was initially thought. 1
Cell-signaling pathways convert a stimulus into cellular action, and they generally have an initiation phase and an effector phase. The initiation phase usually transmits information from a ligand, through interaction with its receptor, to the first biochemical event, resulting in propagation of the pathway. The effector phase executes the biochemical events that result in the desired (often complex) cellular response. In contrast to signaling pathways that use phosphorylation, glycosylation, ubiquitylation or acetylation to alter protein properties, those that require proteolysis are irreversible, because cells are not known to possess a mechanism to re-ligate cut peptide bonds.
Apoptosis is such a pathway; indeed, one can appreciate that once a cell has made the decision to commit suicide, there is not much point in going back. Apoptosis pathways in mammals can have one of two initiation phases, and which pathway is selected depends on the nature of the death signal to be integrated
1. http://darwins-god.blogspot.com.br/search?q=multicellular
2. http://www.nature.com/cdd/journal/v9/n4/full/4400991a.html
Last edited by Otangelo on Sun Feb 28, 2021 1:42 pm; edited 10 times in total