Protons and electrons - and the forces involving them, evidence of fine-tuning, and design
https://reasonandscience.catsboard.com/t3138-protons-and-the-forces-involving-them-evidence-of-fine-tuning-and-design
Proton mass vs electron mass
The proton mass is 1836 times that of an electron. If this ratio were off even slightly, molecules would not form properly. It is also interesting to note that although protons are very different in size and mass, the charges are exactly the same in the opposite degree. If this were not the case, again, molecules necessary to support complex life could not form. The same is true of the electromagnetic coupling constant between protons and electrons - it is very precisely balanced to support complex life.
Andrew Thomas: HIDDEN IN PLAIN SIGHT 7 The Fine-Tuned Universe page 12
The value of ε, the Greek letter epsilon, which represents the amount of energy released by the nuclear fusion reaction inside stars. The main fusion reaction combines hydrogen atoms to form helium atoms. The nucleus of a hydrogen atom is formed of a single proton, whereas the nucleus of a helium atom is formed out of two protons and two neutrons. But the nucleus of a helium atom weighs only 99.3% as much as the two protons and the two neutrons out of which it is made. The remaining 0.7% is released as energy during the fusion reaction, the energy coming from the loss of that small amount of mass according to E=mc 2 . Hence, the value of epsilon is 0.7%, or 0.007. Martin Rees then considers the case if the value of epsilon was 0.006 instead of 0.007. This would be the case if the strong nuclear force holding the protons and neutrons together was slightly weaker. As a result of this weakening of the force, protons could not be bound together during fusion and we would be left with a universe composed entirely of hydrogen. There would be no chemistry in such a universe — and no life. Rees then considers the alternative situation in which epsilon was 0.008 instead of 0.007.This would represent a strengthening of the strong nuclear force. In that case, all the hydrogen would have disappeared from the universe shortly after the Big Bang, quickly fused into helium. There would then have been no fuel for stars, no water, and no life. So, according to Martin Rees: "If epsilon were 0.006 or 0.008, we could not exist." 3
The only plausible forms of advanced life that could originate anywhere in the universe are based on atoms. You might think that the mass of a particle doesn’t really matter that much. The mass of particles is very important in determining their longevity, their suitability in sustaining nuclear reactions in stars, and their suitability for chemistry.
In order to have evidence that life-permitting physics is a small subset among possibilities, we must have some idea of the range of possibilities. There is a maximum mass for particles as set by the Planck scale. The current concept of mass would become incoherent if particles could exceed the so-called Planck mass. The Standard Model provides a means of computing quantum corrections that affect masses, resulting in a natural scale for particle masses. Let’s examine whether or not the mass of certain particles has to be finely tuned to support life.
The Masses of the Electron and the Proton
Stephen Hawking. The Grand Design, p. 160
“If protons were 0.2 percent heavier, they would decay into neutrons, destabilizing atoms.”
1) For hydrogen to exist the mass of an electron must be less than the difference in the masses of a neutron and a proton else the electron would be captured by the proton to form a neutron. Without hydrogen, there would be no water and no long-lived stars (e.g. Helium stars burn out 30 times faster).
2) Atoms are only stable if the radius of an electron orbit is significantly greater than the size of the nucleus – this depends on the ratio of the electron and proton masses such that αβ/αs < 1/1000.
3) The energy scale for chemical reactions should be much smaller than that for nuclear reactions. Otherwise, information could not be stably stored because the type of elements in molecules would be changing because chemical identity would not be maintained. This requires the ratio of the electron and proton masses to be finely tuned such that α2β/αs2 < 1/1000.
4) Unless the fourth root of β is less than about 1/3, molecular structures would be unstable. They would basically be continually melting and thus disrupting the ability to store information.
5) The stability of the proton requires the electromagnetic coupling constant to be less than the difference of the masses of the down quark and up quark divided by a constant. This enables the extra electromagnetic mass-energy of a proton relative to a neutron to be counter-balanced by the bare quark masses.
6) This fine-tuning is related to the electromagnetic coupling constant.
7) Stars will not be stable unless β > 0.01 α2
Note that life-permitting criteria 2-5 and 7 also depend on one or more coupling constants and thus reinforces my previous arguments about the difficulties in getting simultaneous solutions to so many independent equations. There are also some additional constraints on the masses of the proton and electron not necessarily shown in Barnes’s diagram:
A constraint on the main nuclear reaction in stars. This depends on a finely tuned strong nuclear force strength as previously mentioned but also depends on a particular relationship of the masses of the up and down quarks and the electron.
The ratio of the mass of the electron to the proton also affects the ability of stars to output photons at energy levels that break chemical bonds (this was also referenced in my previous blog because it also depended on force strengths). The dashed line in the diagram represents that constraint.
The mass of the electron and proton also show up in the equation for the cosmological parameter Q.
These tight constraints on the life-permitting region for the mass of the electron are even more surprising because the values are deemed “unnaturally low” to begin with. Barnes elucidates this issue: “There are, then, two independent ways in which the masses of the basic constituents of matter are surprisingly small … the Hierarchy Problem … and the flavour problem. … The electron mass is unnaturally smaller than its (unnaturally small) natural scale set by the Higgs condensate.” These are called problems simply because they require fine-tuning – the values they take on are quite different than the natural scale. It’s possible that new physics discoveries might minimize the unnaturalness somewhat but the life-permitting ranges are so tight that there is no basis for assuming that the fine-tuning will go away.
There is also a tight constraint on the charge of the electron. The electromagnetic coupling constant can be expressed in terms of a ratio involving the square of the charge of an electron. Thus, the numerous constraints referenced in my previous blog can also be viewed as a dependence on the charge of the electron. Thus, consider again the fine-tuning necessary for the production of carbon and oxygen in stars. This required fine-tuning of the electromagnetic coupling constant to 1 part in 10,000. Thus, another way of looking at this is that if the electron differed in charge by more than 1 part in 100,000,000 in either direction then the universe would basically be devoid of carbon or oxygen or both.
In order to understand more details about the mass of the proton, a little background will be helpful. A proton is comprised of 2 up quarks and a down quark and a neutron is comprised of 2 down quarks and an up quark. Most of the mass of these composite particles is derived not from the quarks but from the energy due to the strong force that is constraining them. This binding energy is equivalent to mass as per Einstein’s famous equation: E=mc2. Thus, we should also examine the sensitivity of the quark masses.
The Stability of the Proton
Not all types of particles are stable; many of them “decay” or disintegrate after a while into other types of particles. The “half-life” tells how long this typically takes. For example, neutrons have a half-life of about ten minutes. Neutrons usually disintegrate into a proton, an electron, and an anti-neutrino. Fortunately, protons are stable. (At least, it is known that they last much longer than the age of the universe.) That is fortunate because the nucleus of ordinary hydrogen (hydrogen 1) consists of just a proton, and if that were unstable, there would be no ordinary hydrogen in the world. And without hydrogen, there would be no water, no organic molecules, no hydrogen-burning stars like the Sun—in short, no possibility of life as we know it. Why isn’t the instability of the neutron equally disastrous, considering that all nuclei except hydrogen 1 contain neutrons? And, for that matter, how can it be that there are neutrons in all those nuclei, like carbon 12 and oxygen 16, if neutrons do last only ten minutes or so? The answer is that whereas an isolated neutron is unstable, a neutron can be quite stable if it is inside an atomic nucleus with other neutrons and protons. The reason for this has to do with a subtle quantum effect called “Fermi energy.” The fact that isolated neutrons are unstable does not matter very much, since isolated neutrons, having no electric charge, do not bind together with electrons to form atoms. The interesting question is how the universe avoids the disaster—not for it but for us—of protons being unstable.
Why is the proton stable and the neutron unstable?
The key is that the neutron is a tiny bit heavier than the proton. The mass of a neutron is 939.565 MeV, while the mass of a proton is 938.272 MeV— a difference of only a seventh of a percent. Relativity theory tells us that mass is the same thing as energy, so this is the same as saying that a neutron has a little bit more energy packed inside it than a proton does. Because of that, a neutron can decay into a proton plus some other particles, and release energy in the process. But a proton cannot decay into a neutron because it does not have enough energy to do so. Because a neutron has a little more energy inside it than a proton, extra energy would have to be supplied to a proton to get it to turn into a neutron.
If things were the other way, if the proton’s mass were even a fraction of a percent larger than the neutron’s, then neutrons would be stable and protons would be unstable, which means that there would be no hydrogen 1, and we would not be here. The reason for the happy fact that protons are slightly lighter than neutrons has to do with the properties of the quarks out of which protons and neutrons are made. The u quark is slightly lighter than the d quark, and a proton has a preponderance of u quarks in it, while the neutron has a preponderance of d quarks. However, nobody yet knows why u quarks are lighter than d quarks rather than the other way around. God, Luck, or a multiverse. There is no question - God is the best answer. 1
The neutron is slightly heavier than the proton by about 1.293 MeV If the mass of the neutron were increased by another 1.4 MeV—that is, by one part in 700 of its actual mass of about 938 MeV—then one of the key steps by which stars burn their hydrogen to helium could not occur. The main process by which hydrogen is burnt to helium in stars is a proton-proton collision, in which two protons form a coupled system, the diproton while flashing past each other. During that time, the two-proton system can undergo a decay via the weak force to form a deuteron, which is a nucleus containing one proton and one neutron. The conversion takes place by the emission of a positron and an electron neutrino:
p+p → deuteron+positron+electron neutrino+0.42 MeV of energy.
About 1.0 MeV more energy is then released by positron/electron annihilation, making a total energy release of 1.42 MeV. This process can occur because the deuteron is less massive than two protons, even though the neutron itself is more massive. The reason is that the binding energy of the strong force between the proton and neutron in the deuteron is approximately 2.2 MeV, thus overcompensating by about 1 MeV for the greater mass of the neutron. If the neutron’s mass were increased by around 1.42 MeV, however, then neither this reaction nor any other reaction leading to deuterium could proceed, because those reactions would become endothermic instead of exothermic (that is, they would absorb energy instead of producing it). Since it is only via the production of deuterium that hydrogen can be burnt to helium, it follows that, if the mass of the neutron were increased beyond 1.4 MeV, stars could not exist.
On the other hand, a small decrease in the neutron mass of around 0.5 to 0.7 MeV would result in nearly equal numbers of protons and neutrons in the early stages of the Big Bang, since neutrons would move from being energetically disfavored to being energetically favored. The protons and neutrons would then combine to form deuterium and tritium, which would in turn fuse via the strong force to form 4He, resulting in an almost all-helium universe. This would have severe life-inhibiting consequences, since helium stars have a lifetime of at most 300 million years and are much less stable than hydrogen-burning stars, thus providing much less time and stability for the evolution of beings of comparable intelligence to ourselves.
A decrease in the neutron mass beyond 0.8 MeV, however, would result in neutrons becoming energetically favored, along with free protons being converted to neutrons, and hence an initially all-neutron universe. Contrary to what Barrow and Tipler argue, however, it is unclear to what extent, if any, this would have life-inhibiting effects. So the above argument establishes a one-sided fine-tuning of the neutron/ proton mass difference. Since the maximum life-permitting mass difference is 1. 4 MeV, and the mass of the neutron is in the order of 1,000 MeV, the degree of one-sided fine-tuning relative to the neutron mass is at least one part in 700, or less, given that the lower bound of the total theoretically possible range of variation in the neutron mass, R, is in the order of the neutron mass itself—that is, 1,000 MeV. Another plausible lower bound of the theoretically possible range R is given by the range of quark masses. According to the Standard Model of particle physics, the proton is composed of two up quarks and one down quark (uud), whereas the neutron is composed of one up quark and two down quarks (udd). Thus we could define the neutron and proton in terms of their quark constituents. The reason the neutron is heavier than the proton is that the down quark has a mass of l0MeV, which is 4 MeV more than the mass of the up quark. This overcompensates by about 1.3 MeV for the 2.7 MeV contribution of the electric charge of the proton to its mass. (Most of the mass of the proton and neutron, however, is due to gluon exchange between the quarks. The quark masses range from 6 MeV for the up quark to 180,000 MeV for the top quark. Thus a 1.42 MeV increase in the neutron mass —which would correspond to a 1.42 MeV increase in the down quark mass—is only a mere one part in 126,000 of the total range of quark masses, resulting in a lower bound for one-sided fine-tuning of about one part in 126,000 of the range of quark masses. Furthermore, since the down quark mass must be greater than zero, its total life-permitting range is 0 to 11.4 MeV, providing a total two-sided fine-tuning of about one part in 18,000 of the range of quark masses.
The atom itself is a bundle of numerous very fortunate "coincidences". Within the atom, the neutron is just slightly more massive than the proton, which means that free neutrons can decay and turn into protons. A free neutron is unstable and will decay into a proton in about 10 minutes - if not within a nucleus. If the proton were larger and had a tendency to decay rather than the neutron, the very structure of the universe would be impossible. A free proton has a half-life of ~10^33 years. 2
Quark masses that make up atoms are fine-tuned by about 1 part in 18000. The mass of quarks influences tendencies of decay, and if not in the right range, the very structure of the universe would be impossible.
STEPHEN M. BARR: Modern Physics and Ancient Faith page 124
The Stability of the Proton
Not all types of particles are stable; many of them “decay” or disintegrate after a while into other types of particles. The “half-life” tells how long this typically takes. For example, neutrons have a half-life of about ten minutes. Neutrons usually disintegrate into a proton, an electron, and an anti-neutrino. Fortunately, protons are stable. (At least, it is known that they last much longer than the age of the universe.) That is fortunate because the nucleus of ordinary hydrogen (hydrogen 1) consists of just a proton, and if that were unstable, there would be no ordinary hydrogen in the world. And without hydrogen, there would be no water, no organic molecules, no hydrogen-burning stars like the Sun—in short no possibility of life as we know it. Why isn’t the instability of the neutron equally disastrous, considering that all nuclei except hydrogen 1 contain neutrons? And, for that matter, how can it be that there are neutrons in all those nuclei, like carbon 12 and oxygen 16, if neutrons do last only ten minutes or so? The answer is that whereas an isolated neutron is unstable, a neutron can be quite stable if it is inside an atomic nucleus with other neutrons and protons. The reason for this has to do with a subtle quantum effect called “Fermi energy.” The fact that isolated neutrons are unstable does not matter very much, since isolated neutrons, having no electric charge, do not bind together with electrons to form atoms.
The interesting question is how the universe avoids the disaster—not for it but for us—of protons being unstable. Why is the proton stable and the neutron unstable? The key is that the neutron is a tiny bit heavier than the proton. The mass of a neutron is 939.565 MeV, while the mass of a proton is 938.272 MeV— a difference of only a seventh of a percent. Relativity theory tells us that mass is the same thing as energy, so this is the same as saying that a neutron has a little bit more energy packed inside it than a proton does. Because of that, a neutron can decay into a proton plus some other particles, and release energy in the process. But a proton cannot decay into a neutron because it does not have enough energy to do so. Because a neutron has a little more energy inside it than a proton, extra energy would have to be supplied to a proton to get it to turn into a neutron. If things were the other way, if the proton’s mass were even a fraction of a percent larger than the neutron’s, then neutrons would be stable and protons would be unstable, which means that there would be no hydrogen 1, and we would not be here.
The reason for the happy fact that protons are slightly lighter than neutrons has to do with the properties of the quarks out of which protons and neutrons are made. The u quark is slightly lighter than the d quark, and a proton has a preponderance of u quarks in it, while the neutron has a preponderance of d quarks. However, nobody yet knows why u quarks are lighter than d quarks rather than the other way around.
God, Luck, or a multiverse. There is no question - God is the best answer.
1. 1. Modern Physics and Ancient Faith STEPHEN M. BARR page 124
2. http://www.detectingdesign.com/detectingdesign.html
3. HIDDEN IN PLAIN SIGHT 7 The Fine-Tuned Universe
https://reasonandscience.catsboard.com/t3138-protons-and-the-forces-involving-them-evidence-of-fine-tuning-and-design
Proton mass vs electron mass
The proton mass is 1836 times that of an electron. If this ratio were off even slightly, molecules would not form properly. It is also interesting to note that although protons are very different in size and mass, the charges are exactly the same in the opposite degree. If this were not the case, again, molecules necessary to support complex life could not form. The same is true of the electromagnetic coupling constant between protons and electrons - it is very precisely balanced to support complex life.
Andrew Thomas: HIDDEN IN PLAIN SIGHT 7 The Fine-Tuned Universe page 12
The value of ε, the Greek letter epsilon, which represents the amount of energy released by the nuclear fusion reaction inside stars. The main fusion reaction combines hydrogen atoms to form helium atoms. The nucleus of a hydrogen atom is formed of a single proton, whereas the nucleus of a helium atom is formed out of two protons and two neutrons. But the nucleus of a helium atom weighs only 99.3% as much as the two protons and the two neutrons out of which it is made. The remaining 0.7% is released as energy during the fusion reaction, the energy coming from the loss of that small amount of mass according to E=mc 2 . Hence, the value of epsilon is 0.7%, or 0.007. Martin Rees then considers the case if the value of epsilon was 0.006 instead of 0.007. This would be the case if the strong nuclear force holding the protons and neutrons together was slightly weaker. As a result of this weakening of the force, protons could not be bound together during fusion and we would be left with a universe composed entirely of hydrogen. There would be no chemistry in such a universe — and no life. Rees then considers the alternative situation in which epsilon was 0.008 instead of 0.007.This would represent a strengthening of the strong nuclear force. In that case, all the hydrogen would have disappeared from the universe shortly after the Big Bang, quickly fused into helium. There would then have been no fuel for stars, no water, and no life. So, according to Martin Rees: "If epsilon were 0.006 or 0.008, we could not exist." 3
The only plausible forms of advanced life that could originate anywhere in the universe are based on atoms. You might think that the mass of a particle doesn’t really matter that much. The mass of particles is very important in determining their longevity, their suitability in sustaining nuclear reactions in stars, and their suitability for chemistry.
In order to have evidence that life-permitting physics is a small subset among possibilities, we must have some idea of the range of possibilities. There is a maximum mass for particles as set by the Planck scale. The current concept of mass would become incoherent if particles could exceed the so-called Planck mass. The Standard Model provides a means of computing quantum corrections that affect masses, resulting in a natural scale for particle masses. Let’s examine whether or not the mass of certain particles has to be finely tuned to support life.
The Masses of the Electron and the Proton
Stephen Hawking. The Grand Design, p. 160
“If protons were 0.2 percent heavier, they would decay into neutrons, destabilizing atoms.”

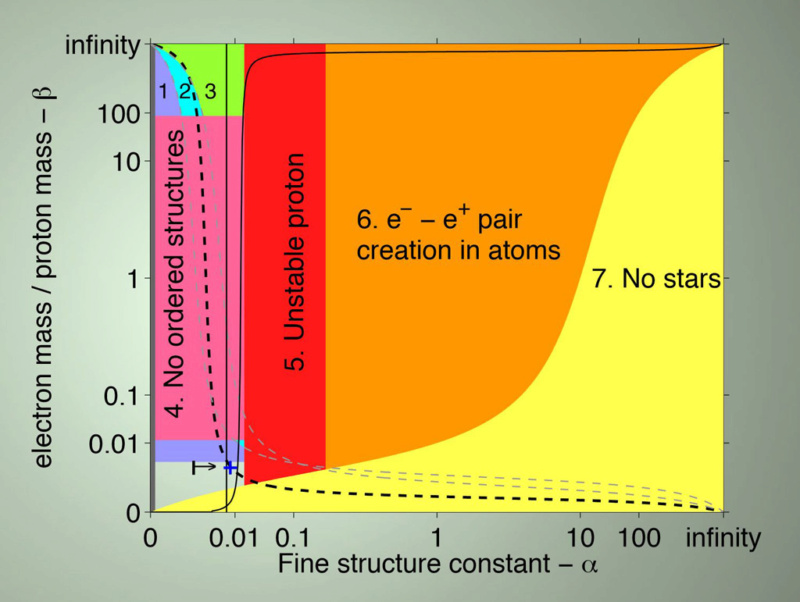
Notes About Diagram: The graph’s axes are scaled based on arctan(log10[x]) such that [0,∞] maps to a finite range. Basically, these are just the dimensionless fundamental constants that convey the strength of the fundamental forces. Here is some notation used in the descriptions of the life-permitting criteria:
α – the electromagnetic coupling constant (also referred to as the fine-structure constant)
αs – the coupling constant for the strong nuclear force
β – the ratio of the mass of an electron to the mass of a proton
The tiny life-permitting region has to simultaneous satisfy each of the following life-permitting criteria and our universe’s values are at the ‘+’ sign near the lower left:1) For hydrogen to exist the mass of an electron must be less than the difference in the masses of a neutron and a proton else the electron would be captured by the proton to form a neutron. Without hydrogen, there would be no water and no long-lived stars (e.g. Helium stars burn out 30 times faster).
2) Atoms are only stable if the radius of an electron orbit is significantly greater than the size of the nucleus – this depends on the ratio of the electron and proton masses such that αβ/αs < 1/1000.
3) The energy scale for chemical reactions should be much smaller than that for nuclear reactions. Otherwise, information could not be stably stored because the type of elements in molecules would be changing because chemical identity would not be maintained. This requires the ratio of the electron and proton masses to be finely tuned such that α2β/αs2 < 1/1000.
4) Unless the fourth root of β is less than about 1/3, molecular structures would be unstable. They would basically be continually melting and thus disrupting the ability to store information.
5) The stability of the proton requires the electromagnetic coupling constant to be less than the difference of the masses of the down quark and up quark divided by a constant. This enables the extra electromagnetic mass-energy of a proton relative to a neutron to be counter-balanced by the bare quark masses.
6) This fine-tuning is related to the electromagnetic coupling constant.
7) Stars will not be stable unless β > 0.01 α2
Note that life-permitting criteria 2-5 and 7 also depend on one or more coupling constants and thus reinforces my previous arguments about the difficulties in getting simultaneous solutions to so many independent equations. There are also some additional constraints on the masses of the proton and electron not necessarily shown in Barnes’s diagram:
A constraint on the main nuclear reaction in stars. This depends on a finely tuned strong nuclear force strength as previously mentioned but also depends on a particular relationship of the masses of the up and down quarks and the electron.
The ratio of the mass of the electron to the proton also affects the ability of stars to output photons at energy levels that break chemical bonds (this was also referenced in my previous blog because it also depended on force strengths). The dashed line in the diagram represents that constraint.
The mass of the electron and proton also show up in the equation for the cosmological parameter Q.
These tight constraints on the life-permitting region for the mass of the electron are even more surprising because the values are deemed “unnaturally low” to begin with. Barnes elucidates this issue: “There are, then, two independent ways in which the masses of the basic constituents of matter are surprisingly small … the Hierarchy Problem … and the flavour problem. … The electron mass is unnaturally smaller than its (unnaturally small) natural scale set by the Higgs condensate.” These are called problems simply because they require fine-tuning – the values they take on are quite different than the natural scale. It’s possible that new physics discoveries might minimize the unnaturalness somewhat but the life-permitting ranges are so tight that there is no basis for assuming that the fine-tuning will go away.
There is also a tight constraint on the charge of the electron. The electromagnetic coupling constant can be expressed in terms of a ratio involving the square of the charge of an electron. Thus, the numerous constraints referenced in my previous blog can also be viewed as a dependence on the charge of the electron. Thus, consider again the fine-tuning necessary for the production of carbon and oxygen in stars. This required fine-tuning of the electromagnetic coupling constant to 1 part in 10,000. Thus, another way of looking at this is that if the electron differed in charge by more than 1 part in 100,000,000 in either direction then the universe would basically be devoid of carbon or oxygen or both.
In order to understand more details about the mass of the proton, a little background will be helpful. A proton is comprised of 2 up quarks and a down quark and a neutron is comprised of 2 down quarks and an up quark. Most of the mass of these composite particles is derived not from the quarks but from the energy due to the strong force that is constraining them. This binding energy is equivalent to mass as per Einstein’s famous equation: E=mc2. Thus, we should also examine the sensitivity of the quark masses.
The Stability of the Proton
Not all types of particles are stable; many of them “decay” or disintegrate after a while into other types of particles. The “half-life” tells how long this typically takes. For example, neutrons have a half-life of about ten minutes. Neutrons usually disintegrate into a proton, an electron, and an anti-neutrino. Fortunately, protons are stable. (At least, it is known that they last much longer than the age of the universe.) That is fortunate because the nucleus of ordinary hydrogen (hydrogen 1) consists of just a proton, and if that were unstable, there would be no ordinary hydrogen in the world. And without hydrogen, there would be no water, no organic molecules, no hydrogen-burning stars like the Sun—in short, no possibility of life as we know it. Why isn’t the instability of the neutron equally disastrous, considering that all nuclei except hydrogen 1 contain neutrons? And, for that matter, how can it be that there are neutrons in all those nuclei, like carbon 12 and oxygen 16, if neutrons do last only ten minutes or so? The answer is that whereas an isolated neutron is unstable, a neutron can be quite stable if it is inside an atomic nucleus with other neutrons and protons. The reason for this has to do with a subtle quantum effect called “Fermi energy.” The fact that isolated neutrons are unstable does not matter very much, since isolated neutrons, having no electric charge, do not bind together with electrons to form atoms. The interesting question is how the universe avoids the disaster—not for it but for us—of protons being unstable.
Why is the proton stable and the neutron unstable?
The key is that the neutron is a tiny bit heavier than the proton. The mass of a neutron is 939.565 MeV, while the mass of a proton is 938.272 MeV— a difference of only a seventh of a percent. Relativity theory tells us that mass is the same thing as energy, so this is the same as saying that a neutron has a little bit more energy packed inside it than a proton does. Because of that, a neutron can decay into a proton plus some other particles, and release energy in the process. But a proton cannot decay into a neutron because it does not have enough energy to do so. Because a neutron has a little more energy inside it than a proton, extra energy would have to be supplied to a proton to get it to turn into a neutron.
If things were the other way, if the proton’s mass were even a fraction of a percent larger than the neutron’s, then neutrons would be stable and protons would be unstable, which means that there would be no hydrogen 1, and we would not be here. The reason for the happy fact that protons are slightly lighter than neutrons has to do with the properties of the quarks out of which protons and neutrons are made. The u quark is slightly lighter than the d quark, and a proton has a preponderance of u quarks in it, while the neutron has a preponderance of d quarks. However, nobody yet knows why u quarks are lighter than d quarks rather than the other way around. God, Luck, or a multiverse. There is no question - God is the best answer. 1
The neutron is slightly heavier than the proton by about 1.293 MeV If the mass of the neutron were increased by another 1.4 MeV—that is, by one part in 700 of its actual mass of about 938 MeV—then one of the key steps by which stars burn their hydrogen to helium could not occur. The main process by which hydrogen is burnt to helium in stars is a proton-proton collision, in which two protons form a coupled system, the diproton while flashing past each other. During that time, the two-proton system can undergo a decay via the weak force to form a deuteron, which is a nucleus containing one proton and one neutron. The conversion takes place by the emission of a positron and an electron neutrino:
p+p → deuteron+positron+electron neutrino+0.42 MeV of energy.
About 1.0 MeV more energy is then released by positron/electron annihilation, making a total energy release of 1.42 MeV. This process can occur because the deuteron is less massive than two protons, even though the neutron itself is more massive. The reason is that the binding energy of the strong force between the proton and neutron in the deuteron is approximately 2.2 MeV, thus overcompensating by about 1 MeV for the greater mass of the neutron. If the neutron’s mass were increased by around 1.42 MeV, however, then neither this reaction nor any other reaction leading to deuterium could proceed, because those reactions would become endothermic instead of exothermic (that is, they would absorb energy instead of producing it). Since it is only via the production of deuterium that hydrogen can be burnt to helium, it follows that, if the mass of the neutron were increased beyond 1.4 MeV, stars could not exist.
On the other hand, a small decrease in the neutron mass of around 0.5 to 0.7 MeV would result in nearly equal numbers of protons and neutrons in the early stages of the Big Bang, since neutrons would move from being energetically disfavored to being energetically favored. The protons and neutrons would then combine to form deuterium and tritium, which would in turn fuse via the strong force to form 4He, resulting in an almost all-helium universe. This would have severe life-inhibiting consequences, since helium stars have a lifetime of at most 300 million years and are much less stable than hydrogen-burning stars, thus providing much less time and stability for the evolution of beings of comparable intelligence to ourselves.
A decrease in the neutron mass beyond 0.8 MeV, however, would result in neutrons becoming energetically favored, along with free protons being converted to neutrons, and hence an initially all-neutron universe. Contrary to what Barrow and Tipler argue, however, it is unclear to what extent, if any, this would have life-inhibiting effects. So the above argument establishes a one-sided fine-tuning of the neutron/ proton mass difference. Since the maximum life-permitting mass difference is 1. 4 MeV, and the mass of the neutron is in the order of 1,000 MeV, the degree of one-sided fine-tuning relative to the neutron mass is at least one part in 700, or less, given that the lower bound of the total theoretically possible range of variation in the neutron mass, R, is in the order of the neutron mass itself—that is, 1,000 MeV. Another plausible lower bound of the theoretically possible range R is given by the range of quark masses. According to the Standard Model of particle physics, the proton is composed of two up quarks and one down quark (uud), whereas the neutron is composed of one up quark and two down quarks (udd). Thus we could define the neutron and proton in terms of their quark constituents. The reason the neutron is heavier than the proton is that the down quark has a mass of l0MeV, which is 4 MeV more than the mass of the up quark. This overcompensates by about 1.3 MeV for the 2.7 MeV contribution of the electric charge of the proton to its mass. (Most of the mass of the proton and neutron, however, is due to gluon exchange between the quarks. The quark masses range from 6 MeV for the up quark to 180,000 MeV for the top quark. Thus a 1.42 MeV increase in the neutron mass —which would correspond to a 1.42 MeV increase in the down quark mass—is only a mere one part in 126,000 of the total range of quark masses, resulting in a lower bound for one-sided fine-tuning of about one part in 126,000 of the range of quark masses. Furthermore, since the down quark mass must be greater than zero, its total life-permitting range is 0 to 11.4 MeV, providing a total two-sided fine-tuning of about one part in 18,000 of the range of quark masses.
The atom itself is a bundle of numerous very fortunate "coincidences". Within the atom, the neutron is just slightly more massive than the proton, which means that free neutrons can decay and turn into protons. A free neutron is unstable and will decay into a proton in about 10 minutes - if not within a nucleus. If the proton were larger and had a tendency to decay rather than the neutron, the very structure of the universe would be impossible. A free proton has a half-life of ~10^33 years. 2
Quark masses that make up atoms are fine-tuned by about 1 part in 18000. The mass of quarks influences tendencies of decay, and if not in the right range, the very structure of the universe would be impossible.
STEPHEN M. BARR: Modern Physics and Ancient Faith page 124
The Stability of the Proton
Not all types of particles are stable; many of them “decay” or disintegrate after a while into other types of particles. The “half-life” tells how long this typically takes. For example, neutrons have a half-life of about ten minutes. Neutrons usually disintegrate into a proton, an electron, and an anti-neutrino. Fortunately, protons are stable. (At least, it is known that they last much longer than the age of the universe.) That is fortunate because the nucleus of ordinary hydrogen (hydrogen 1) consists of just a proton, and if that were unstable, there would be no ordinary hydrogen in the world. And without hydrogen, there would be no water, no organic molecules, no hydrogen-burning stars like the Sun—in short no possibility of life as we know it. Why isn’t the instability of the neutron equally disastrous, considering that all nuclei except hydrogen 1 contain neutrons? And, for that matter, how can it be that there are neutrons in all those nuclei, like carbon 12 and oxygen 16, if neutrons do last only ten minutes or so? The answer is that whereas an isolated neutron is unstable, a neutron can be quite stable if it is inside an atomic nucleus with other neutrons and protons. The reason for this has to do with a subtle quantum effect called “Fermi energy.” The fact that isolated neutrons are unstable does not matter very much, since isolated neutrons, having no electric charge, do not bind together with electrons to form atoms.
The interesting question is how the universe avoids the disaster—not for it but for us—of protons being unstable. Why is the proton stable and the neutron unstable? The key is that the neutron is a tiny bit heavier than the proton. The mass of a neutron is 939.565 MeV, while the mass of a proton is 938.272 MeV— a difference of only a seventh of a percent. Relativity theory tells us that mass is the same thing as energy, so this is the same as saying that a neutron has a little bit more energy packed inside it than a proton does. Because of that, a neutron can decay into a proton plus some other particles, and release energy in the process. But a proton cannot decay into a neutron because it does not have enough energy to do so. Because a neutron has a little more energy inside it than a proton, extra energy would have to be supplied to a proton to get it to turn into a neutron. If things were the other way, if the proton’s mass were even a fraction of a percent larger than the neutron’s, then neutrons would be stable and protons would be unstable, which means that there would be no hydrogen 1, and we would not be here.
The reason for the happy fact that protons are slightly lighter than neutrons has to do with the properties of the quarks out of which protons and neutrons are made. The u quark is slightly lighter than the d quark, and a proton has a preponderance of u quarks in it, while the neutron has a preponderance of d quarks. However, nobody yet knows why u quarks are lighter than d quarks rather than the other way around.
God, Luck, or a multiverse. There is no question - God is the best answer.
1. 1. Modern Physics and Ancient Faith STEPHEN M. BARR page 124
2. http://www.detectingdesign.com/detectingdesign.html
3. HIDDEN IN PLAIN SIGHT 7 The Fine-Tuned Universe

