Evidence shows that life depends on the right order and arrangement even of atoms !!
https://reasonandscience.catsboard.com/t2975-evidence-shows-that-life-depends-on-the-right-order-and-arrangement-even-of-atoms
This was my introduction discourse in my debate with Leophilius:
Intelligent Design in Abiogenesis? DEBATE | Otangelo Vs Leophilius
https://www.youtube.com/watch?v=eb1YWZz4-d0&lc=z23zub5o0sznuvfyn04t1aokgo0f1ajzwhxo0vjnxnxpbk0h00410.1594768575517304
The origin of life is widely regarded as one of the most difficult open problems in science. ‘Bottom-up’ approaches in the laboratory have not generated anything nearly as complex as a living cell. And what has been achieved, is a far cry from the complexity of anything living. The total lack of any kind of experimental evidence leading to the re-creation of life; not to mention the spontaneous emergence of life… undermines the worldview of who wants materialism to be true. But of course, there is always an excuse: Science is working on it. But is it really justified to put hope that one day a materialistic explanation will be found?
David Denton stated:
We now know not only of the existence of a break between the living and non-living world but also that it represents the most dramatic and fundamental of all the discontinuities of nature. Between a living cell and the most highly ordered non-biological systems, such as a crystal or a snowflake, there is a chasm as vast and absolute as it is possible to conceive.
And Lynn Margulis stated: To go from a bacterium to people is less of a step than to go from a mixture of amino acids to a bacterium.
And Eugene Koonin advisory editorial board of Trends in Genetics stated:
A succession of exceedingly unlikely steps is essential for the origin of life, from the synthesis and accumulation of nucleotides to the origin of translation; through the multiplication of probabilities, these make the final outcome seem almost like a miracle. The difficulties remain formidable. For all the effort, we do not currently have coherent and plausible models for the path from simple organic molecules to the first life forms. Most damningly, the powerful mechanisms of biological evolution were not available for all the stages preceding the emergence of replicator systems. Given all these major difficulties, it appears prudent to seriously consider radical alternatives for the origin of life. "
And in fact, there are basically just two options to consider: Either life emerged by a lucky accident, spontaneously through self-organization by unguided natural events, or through the direct intervention, creative force, and activity of an intelligent designer. Evolution is not a possible explanation, because evolution depends on DNA replication. Many have claimed that physical necessity could have promoted chemical reactions, which eventually resulted in the emergence of life. The problem here however is, that the genetic sequence that specifies the arrangement of proteins can be of any order, there is no constraint by physical needs.
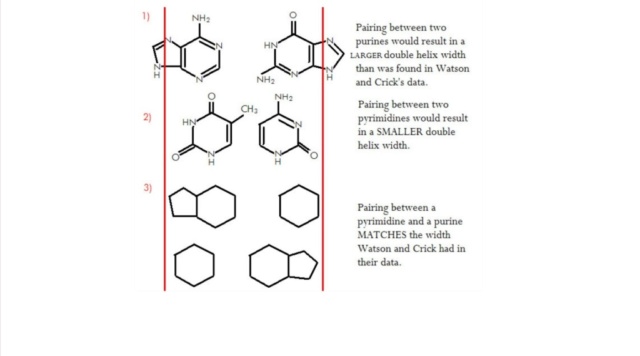
To understand why random events are not a good explanation, we best have a look at the deepest level, on an atomic scale. Life uses just five nucleobases to make DNA and RNA. Two purines, and three pyrimidines. Purines use two rings with nine atoms, pyrimidines use just one ring with six atoms. Hydrogen bonding between purine and pyrimidine bases is fundamental to the biological functions of nucleic acids, as in the formation of the double-helix structure of DNA. This bonding depends on the selection of the right atoms in the ring structure. Pyrimidine rings consist of six atoms: 4 carbon atoms and 2 nitrogen atoms. Purines have nine atoms forming the ring: 5 carbon atoms and 4 nitrogen atoms.
Remarkably, it is the composition of these atoms that permit that the strength of the hydrogen bond that permits to join the two DNA strands and form Watson–Crick base-pairing, and well-known DNA ladder. Neither transcription nor translation of the messages encoded in RNA and DNA would be possible if the strength of the bonds had different values. Hence, life, as we understand it today, would not have arisen.
Now, someone could say, that there could be no different composition, and physical constraints and necessity could eventually permit only this specific order and arrangement of the atoms. Now, in a recent science paper from 2019, Scientists explored how many different chemical arrangements of the atoms to make these nucleobases would be possible. Surprisingly, they found well over a million variants. The remarkable thing is, among the incredible variety of organisms on Earth, these two molecules are essentially the only ones used in life. Why? Are these the only nucleotides that could perform the function of information storage? If not, are they perhaps the best? One might expect that molecules with smaller connected Carbon components should be easier for abiotic chemistry to explore.
According to their scientific analysis, the natural ribosides and deoxyribosides inhabit a fairly redundant ( in other words, superfluous, unnecessary, needless, and nonminimal region of this space. This is a remarkable find and implicitly leads to design. There would be no reason why random events would generate complex, rather than simple, and minimal carbon arrangements. Nor is there physical necessity that says that the composition should be so. This is evidence that a directing intelligent agency is the most plausible explanation. The chemistry space is far too vast to select by chance the right finely-tuned functional life-bearing arrangement.
In the mentioned paper, the investigators asked if other, perhaps equally good, or even better genetic systems would be possible. Their chemical experimentations and studies concluded that the answer is no. Many nearly as good, some equally good, and a few stronger base-pairing analog systems are known. There is no reason why these structures could or would have emerged in this functional complex configuration by random trial and error. There is a complete lack of scientific-materialistic explanations despite decades of attempts to solve the riddle.
What we can see is, that direct intervention, a creative force, the activity of an intelligent agency, a powerful creator, is capable to have the intention and implement the right arrangement of every single atom into functional structures and molecules in a repetitive manner, in the case of DNA, at least 500 thousand nucleotides to store the information to kick-start life, exclusively with four bases, to produce a storage device that uses a genetic code, to store functional, instructional, complex information, functional amino acids, and phospholipids to make membranes, and ultimately, life. Lucky accidents, the spontaneous self-organization by unguided coincidental events, that drove atoms into self-organization in an orderly manner without external direction, chemical non-biological are incapable and unspecific to arrange atoms into the right order to produce the four classes of building blocks, used in all life forms.
Intelligent design advocates commonly point to the intrinsic order of molecules required to permit life the first go. Like the order of amino acids, arranged to permit polypeptides to fold into 3D arrangements that become functional. But, in the following examples, I will demonstrate, that the precise order is required down even on a deeper level of the precise finely adjusted and tuned arrangement of atoms.
According to an estimate made by engineers at Washington University, there are around 10^14 atoms in a typical human cell. Another way of looking at it is that this is 100,000,000,000,000 or 100 trillion atoms. . 1
A human cell hosts about 2 billion proteins. 2
Let's give a closer look to just one protein. Aspartate Carbamoyltransferase, one of the essential enzymes for the synthesis of DNA bases:
The active site of the enzyme is located where two individual catalytic subunits touch, so the position of the two subunits relative to one another is critical. Take just a moment to ponder the immensity of this enzyme. [/color]The entire complex is composed of over 40,000 atoms, each of which plays a vital role. The handful of atoms that actually perform the chemical reaction are the central players. But they are not the only important atoms within the enzyme--every atom plays a supporting pan. The atoms lining the surfaces between subunits are chosen to complement one another exactly, to orchestrate the shifting regulatory motions. The atoms covering the surface are carefully picked to interact optimally with water, ensuring that the enzyme doesn't form a pasty aggregate, but remains an individual, floating factory.
My comment: This is evidence, that the precise order to make life possible goes down to a prerequisite to arrange even atoms in the right order. The following will corroborate this even further !!
Fine-tuning in biochemistry is represented by the strength of the chemical bonds that makes the universal genetic code possible. Neither transcription nor translation of the messages encoded in RNA and DNA would be possible if the strength of the bonds had different values. Hence, life, as we understand it today, would not have arisen. 3
As it happens, the average bond energy of a carbon–oxygen double bond is about 30 kcal per mol higher than that of a carbon–carbon or carbon–nitrogen double bond. If the difference between the average bond energy of a carbon–oxygen double bond and that of a carbon–carbon and carbon–nitrogen double bond were smaller by a few kcal per mol, then the nucleobases guanine, cytosine, and thymine would exist as “enols” and not as “ketones,” and Watson–Crick base-pairing would not exist – nor would the kind of life we know.
My comment: That means, the atom composition of the DNA nucleobases most be of the right atoms to guarantee the right hydrogen bond forces that permit Watson-Crick base-pairing.
Scientists recently explored the "chemical neighborhood" of nucleic acid analogs. Surprisingly, they found well over a million variants 1 Are DNA and RNA the only way to store this information? Or are they perhaps the best way? "There are two kinds of nucleic acids in biology ( RNA and DNA). The remarkable thing is, among the incredible variety of organisms on Earth, these two molecules are essentially the only ones used in life. Why ?.Are these the only molecules that could perform this function? If not, are they perhaps the best? Chemical space is, in principle, unlimited as at least one more atom can almost always be added to any given structure. 2 Any given structure space is limited by the input definitions, as well as by defined structural constraints. One might expect that molecules with smaller connected Carbon components should be easier for abiotic chemistry to explore. According to scientific analysis, the natural ribosides and deoxyribosides inhabit a fairly redundant ( superfluous, unnecessary, needless) and nonminimal region of this space.
My comment: This is a remarkable find and implicitly leads to design. There would be no reason why stochastic events would generate complex, rather than simple, and minimal carbon arrangements. Nor is there physical necessity that says that the arrangement should be so. This is evidence that directing intelligent agency is the most plausible explanation. The chemistry space is far too vast to select by chance the right finely-tuned functional arrangement.
How can it be best explained, that life incorporates a solution to the need for information storage? A large number of possible motifs, both those which have thus-far been synthesized, which represent a minuscule fraction of the possible ones , argue that solutions in this chemical space are unlikely due to unguided stochastic natural events but rather the guiding hand of an intelligent designer.
From the standpoint of xeno and synthetic biology, could other, perhaps equally good, or even better genetic systems be devised? The answer to this question will require sophisticated and protracted chemical experimentation.Studies to date suggest that the answer could be no[/b]. Many nearly as good, some equally good, and a few stronger base-pairing analog systems are known.
My comment: Amazing!! RNA and DNA are probably an OPTIMALLY DESIGNED genetic system. This is clear evidence of design on an atomic level !! DNA molecules are asymmetrical, such property is essential in the processes of DNA replication and transcription. Bases need to be paired between pyrimidines and purines. In molecular biology, complementarity describes a relationship between two structures each following the lock-and-key principle.
Complementarity on an atomic level is the base principle of DNA replication and transcription as it is a property shared between two DNA or RNA sequences, such that when they are aligned antiparallel to each other, the nucleotide bases at each position in the sequences will be complementary, much like looking in the mirror and seeing the reverse of things. This complimentary base pairing is essential for cells to copy information from one generation to another. There is no reason why these structures could or would have emerged in this functional complex configuration by random trial and error processes. There is a complete lack of scientific-materialistic explanations despite decades of attempts to solve the riddle.
Another interesting observation is that RNA and DNA use a five-membered ribose ring structure as a backbone element. It is found that six-membered ring with backbones containing six carbons per sugar unit instead of five carbons and six-membered pyranose rings instead of five-membered furanose rings do not possess the capability of efficient informational Watson–Crick base-pairing.
https://sci-hub.ren/https://pubs.acs.org/doi/10.1021/acs.jcim.9b00632

https://reasonandscience.catsboard.com/t2975-evidence-shows-that-life-depends-on-the-right-order-and-arrangement-even-of-atoms
This was my introduction discourse in my debate with Leophilius:
Intelligent Design in Abiogenesis? DEBATE | Otangelo Vs Leophilius
https://www.youtube.com/watch?v=eb1YWZz4-d0&lc=z23zub5o0sznuvfyn04t1aokgo0f1ajzwhxo0vjnxnxpbk0h00410.1594768575517304
The origin of life is widely regarded as one of the most difficult open problems in science. ‘Bottom-up’ approaches in the laboratory have not generated anything nearly as complex as a living cell. And what has been achieved, is a far cry from the complexity of anything living. The total lack of any kind of experimental evidence leading to the re-creation of life; not to mention the spontaneous emergence of life… undermines the worldview of who wants materialism to be true. But of course, there is always an excuse: Science is working on it. But is it really justified to put hope that one day a materialistic explanation will be found?
David Denton stated:
We now know not only of the existence of a break between the living and non-living world but also that it represents the most dramatic and fundamental of all the discontinuities of nature. Between a living cell and the most highly ordered non-biological systems, such as a crystal or a snowflake, there is a chasm as vast and absolute as it is possible to conceive.
And Lynn Margulis stated: To go from a bacterium to people is less of a step than to go from a mixture of amino acids to a bacterium.
And Eugene Koonin advisory editorial board of Trends in Genetics stated:
A succession of exceedingly unlikely steps is essential for the origin of life, from the synthesis and accumulation of nucleotides to the origin of translation; through the multiplication of probabilities, these make the final outcome seem almost like a miracle. The difficulties remain formidable. For all the effort, we do not currently have coherent and plausible models for the path from simple organic molecules to the first life forms. Most damningly, the powerful mechanisms of biological evolution were not available for all the stages preceding the emergence of replicator systems. Given all these major difficulties, it appears prudent to seriously consider radical alternatives for the origin of life. "
And in fact, there are basically just two options to consider: Either life emerged by a lucky accident, spontaneously through self-organization by unguided natural events, or through the direct intervention, creative force, and activity of an intelligent designer. Evolution is not a possible explanation, because evolution depends on DNA replication. Many have claimed that physical necessity could have promoted chemical reactions, which eventually resulted in the emergence of life. The problem here however is, that the genetic sequence that specifies the arrangement of proteins can be of any order, there is no constraint by physical needs.
To understand why random events are not a good explanation, we best have a look at the deepest level, on an atomic scale. Life uses just five nucleobases to make DNA and RNA. Two purines, and three pyrimidines. Purines use two rings with nine atoms, pyrimidines use just one ring with six atoms. Hydrogen bonding between purine and pyrimidine bases is fundamental to the biological functions of nucleic acids, as in the formation of the double-helix structure of DNA. This bonding depends on the selection of the right atoms in the ring structure. Pyrimidine rings consist of six atoms: 4 carbon atoms and 2 nitrogen atoms. Purines have nine atoms forming the ring: 5 carbon atoms and 4 nitrogen atoms.
Remarkably, it is the composition of these atoms that permit that the strength of the hydrogen bond that permits to join the two DNA strands and form Watson–Crick base-pairing, and well-known DNA ladder. Neither transcription nor translation of the messages encoded in RNA and DNA would be possible if the strength of the bonds had different values. Hence, life, as we understand it today, would not have arisen.
Now, someone could say, that there could be no different composition, and physical constraints and necessity could eventually permit only this specific order and arrangement of the atoms. Now, in a recent science paper from 2019, Scientists explored how many different chemical arrangements of the atoms to make these nucleobases would be possible. Surprisingly, they found well over a million variants. The remarkable thing is, among the incredible variety of organisms on Earth, these two molecules are essentially the only ones used in life. Why? Are these the only nucleotides that could perform the function of information storage? If not, are they perhaps the best? One might expect that molecules with smaller connected Carbon components should be easier for abiotic chemistry to explore.
According to their scientific analysis, the natural ribosides and deoxyribosides inhabit a fairly redundant ( in other words, superfluous, unnecessary, needless, and nonminimal region of this space. This is a remarkable find and implicitly leads to design. There would be no reason why random events would generate complex, rather than simple, and minimal carbon arrangements. Nor is there physical necessity that says that the composition should be so. This is evidence that a directing intelligent agency is the most plausible explanation. The chemistry space is far too vast to select by chance the right finely-tuned functional life-bearing arrangement.
In the mentioned paper, the investigators asked if other, perhaps equally good, or even better genetic systems would be possible. Their chemical experimentations and studies concluded that the answer is no. Many nearly as good, some equally good, and a few stronger base-pairing analog systems are known. There is no reason why these structures could or would have emerged in this functional complex configuration by random trial and error. There is a complete lack of scientific-materialistic explanations despite decades of attempts to solve the riddle.
What we can see is, that direct intervention, a creative force, the activity of an intelligent agency, a powerful creator, is capable to have the intention and implement the right arrangement of every single atom into functional structures and molecules in a repetitive manner, in the case of DNA, at least 500 thousand nucleotides to store the information to kick-start life, exclusively with four bases, to produce a storage device that uses a genetic code, to store functional, instructional, complex information, functional amino acids, and phospholipids to make membranes, and ultimately, life. Lucky accidents, the spontaneous self-organization by unguided coincidental events, that drove atoms into self-organization in an orderly manner without external direction, chemical non-biological are incapable and unspecific to arrange atoms into the right order to produce the four classes of building blocks, used in all life forms.
Intelligent design advocates commonly point to the intrinsic order of molecules required to permit life the first go. Like the order of amino acids, arranged to permit polypeptides to fold into 3D arrangements that become functional. But, in the following examples, I will demonstrate, that the precise order is required down even on a deeper level of the precise finely adjusted and tuned arrangement of atoms.
According to an estimate made by engineers at Washington University, there are around 10^14 atoms in a typical human cell. Another way of looking at it is that this is 100,000,000,000,000 or 100 trillion atoms. . 1
A human cell hosts about 2 billion proteins. 2
Let's give a closer look to just one protein. Aspartate Carbamoyltransferase, one of the essential enzymes for the synthesis of DNA bases:
The active site of the enzyme is located where two individual catalytic subunits touch, so the position of the two subunits relative to one another is critical. Take just a moment to ponder the immensity of this enzyme. [/color]The entire complex is composed of over 40,000 atoms, each of which plays a vital role. The handful of atoms that actually perform the chemical reaction are the central players. But they are not the only important atoms within the enzyme--every atom plays a supporting pan. The atoms lining the surfaces between subunits are chosen to complement one another exactly, to orchestrate the shifting regulatory motions. The atoms covering the surface are carefully picked to interact optimally with water, ensuring that the enzyme doesn't form a pasty aggregate, but remains an individual, floating factory.
My comment: This is evidence, that the precise order to make life possible goes down to a prerequisite to arrange even atoms in the right order. The following will corroborate this even further !!
Fine-tuning in biochemistry is represented by the strength of the chemical bonds that makes the universal genetic code possible. Neither transcription nor translation of the messages encoded in RNA and DNA would be possible if the strength of the bonds had different values. Hence, life, as we understand it today, would not have arisen. 3
As it happens, the average bond energy of a carbon–oxygen double bond is about 30 kcal per mol higher than that of a carbon–carbon or carbon–nitrogen double bond. If the difference between the average bond energy of a carbon–oxygen double bond and that of a carbon–carbon and carbon–nitrogen double bond were smaller by a few kcal per mol, then the nucleobases guanine, cytosine, and thymine would exist as “enols” and not as “ketones,” and Watson–Crick base-pairing would not exist – nor would the kind of life we know.
My comment: That means, the atom composition of the DNA nucleobases most be of the right atoms to guarantee the right hydrogen bond forces that permit Watson-Crick base-pairing.
Scientists recently explored the "chemical neighborhood" of nucleic acid analogs. Surprisingly, they found well over a million variants 1 Are DNA and RNA the only way to store this information? Or are they perhaps the best way? "There are two kinds of nucleic acids in biology ( RNA and DNA). The remarkable thing is, among the incredible variety of organisms on Earth, these two molecules are essentially the only ones used in life. Why ?.Are these the only molecules that could perform this function? If not, are they perhaps the best? Chemical space is, in principle, unlimited as at least one more atom can almost always be added to any given structure. 2 Any given structure space is limited by the input definitions, as well as by defined structural constraints. One might expect that molecules with smaller connected Carbon components should be easier for abiotic chemistry to explore. According to scientific analysis, the natural ribosides and deoxyribosides inhabit a fairly redundant ( superfluous, unnecessary, needless) and nonminimal region of this space.
My comment: This is a remarkable find and implicitly leads to design. There would be no reason why stochastic events would generate complex, rather than simple, and minimal carbon arrangements. Nor is there physical necessity that says that the arrangement should be so. This is evidence that directing intelligent agency is the most plausible explanation. The chemistry space is far too vast to select by chance the right finely-tuned functional arrangement.
How can it be best explained, that life incorporates a solution to the need for information storage? A large number of possible motifs, both those which have thus-far been synthesized, which represent a minuscule fraction of the possible ones , argue that solutions in this chemical space are unlikely due to unguided stochastic natural events but rather the guiding hand of an intelligent designer.
From the standpoint of xeno and synthetic biology, could other, perhaps equally good, or even better genetic systems be devised? The answer to this question will require sophisticated and protracted chemical experimentation.Studies to date suggest that the answer could be no[/b]. Many nearly as good, some equally good, and a few stronger base-pairing analog systems are known.
My comment: Amazing!! RNA and DNA are probably an OPTIMALLY DESIGNED genetic system. This is clear evidence of design on an atomic level !! DNA molecules are asymmetrical, such property is essential in the processes of DNA replication and transcription. Bases need to be paired between pyrimidines and purines. In molecular biology, complementarity describes a relationship between two structures each following the lock-and-key principle.
Complementarity on an atomic level is the base principle of DNA replication and transcription as it is a property shared between two DNA or RNA sequences, such that when they are aligned antiparallel to each other, the nucleotide bases at each position in the sequences will be complementary, much like looking in the mirror and seeing the reverse of things. This complimentary base pairing is essential for cells to copy information from one generation to another. There is no reason why these structures could or would have emerged in this functional complex configuration by random trial and error processes. There is a complete lack of scientific-materialistic explanations despite decades of attempts to solve the riddle.
Another interesting observation is that RNA and DNA use a five-membered ribose ring structure as a backbone element. It is found that six-membered ring with backbones containing six carbons per sugar unit instead of five carbons and six-membered pyranose rings instead of five-membered furanose rings do not possess the capability of efficient informational Watson–Crick base-pairing.
https://sci-hub.ren/https://pubs.acs.org/doi/10.1021/acs.jcim.9b00632



