https://reasonandscience.catsboard.com/t2285-iron-sulfur-clusters-basic-building-blocks-for-life
The Last Universal Common Ancestors (LUCA's) biochemistry was replete with FeS clusters and radical reaction mechanisms. 14 Proteins containing Fe–S clusters exist in all living organisms and play an essential role in diverse biological processes at the cellular level. It was thought for a long time that the assembly of Fe–S proteins occurred spontaneously since the process was easily replicated chemically in vitro and led to the view that these cofactors can assemble spontaneously in proteins. However, genetic, biochemical, molecular, and cell biology studies in the late 1990s provided ample evidence that demonstrated that the assembly of Fe–S clusters in vivo is a catalyzed process rather than a spontaneous one and that it requires a plethora of genes assisting in the maturation of Fe–S clusters and their insertion into the apoproteins. Therefore, the formation of intracellular Fe–S clusters does not occur spontaneously but requires a complex biosynthetic machinery. Despite the relative simplicity of Fe–S clusters in terms of structure and composition, their synthesis, assembly, and transfer to apoproteins is a highly complex and coordinated process in living cells. Numerous Fe–S cluster synthesis components have been discovered that assist Fe–S protein maturation according to distinct biosynthetic principles in several model organisms 15



The genes that participate in Fe–S cluster synthesis appears to be conserved in bacteria, fungi, animals, and plants. High sequence identity or similarity is indicated by ++ and +, limited sequence similarity is depicted by ?, and no
evident similarity by −.
With the availability of the whole-genome sequencing database, it was revealed that the Fe–S biogenesis machinery is widespread and highly conserved from prokaryotes and eukaryotes. Three distinct Fe–S protein biogenesis systems have been identified in prokaryotes so far: the NIF system present in nitrogen-fixing bacteria (A. vinelandii) is specialized in the assembly of the complex Fe–S protein nitrogenase, which is responsible for the conversion of N2 to NH3 in nitrogen-fixing bacteria; the ISC system is responsible for the generation of the majority of cellular Fe–S proteins and thus, might perform a general housekeeping biosynthetic function particularly under normal and low oxygen concentrations. Finally, the SUF (sulfur-mobilization) machinery was discovered as an independent assemblage that might be used preferentially under oxidative stress and iron-limiting conditions.
Sulfur is an essential element, being a constituent of many proteins and cofactors. Iron-sulfur (FeS) centers are essential protein cofactors in all forms of life. Various biosynthetic pathways were found to be tightly interconnected through complex crosstalk mechanisms that crucially depend on the bio-availability of the metal ions iron, molybdenum, tungsten, nickel, copper, and zinc. Proteins requiring Fe/S clusters in their active site have been localized in mitochondria, cytosol and nucleus where they are involved in rather diverse functions such as the TCA cycle, amino acid biosynthesis, bacterial and mitochondrial respiration, co-factor biosynthesis, ribosome assembly, regulation of protein translation, DNA replication and DNA repair. Hence the process of iron-sulphur biosynthesis is essential to almost all forms of life.
The prevalence of these proteins on the metabolic pathways of most organisms leads some scientists to theorize that iron–sulfur compounds had a significant role in the origin of life in the iron–sulfur world theory. The iron–sulfur world hypothesis is a set of proposals for the origin of life and the early evolution of life advanced in a series of articles between 1988 and 1992 by Günter Wächtershäuser. FeS cluster assembly is a complex process involving the mobilisation of Fe and S atoms from storage sources, their assembly into [Fe-S] form, their transport to specific cellular locations, and their transfer to recipient apoproteins. These ancient and essential components of the cell machinery depend on ferrous iron and sulfur, elements that are readily available in a reducing atmosphere, but are scarce in an oxygen-rich atmosphere; that reactive oxygen species generated by aerobic respiration are damaging to FeS clusters; that free iron and sulfide released by FeS clusters are toxic to cells; that therefore complex mechanisms are needed to coordinate the synthesis of these simple cofactors, and that these pathways have to be compartmentalized in organelles of prokaryotic origin.
That adds further problems, if the prebiotic atmosphere was oxygen rich, and not reducing, as evidences suggests. Fe-S clusters are partners in the origin of life that predate cells, acetyl-CoA metabolism, DNA, and the RNA world.13 Nar1 is a essential component of a cytosolic Fe/S protein assembly machinery. Required for maturation of extramitochondrial Fe/S proteins. 12 Thus, Nar1 is both a target and a component of the cellular Fe/S protein biogenesis machinery creating an interesting “chicken and egg” situation for its maturation process Conserved Iron–Sulfur (Fe–S) clusters are found in a growing family of metalloproteins that are implicated in prokaryotic and eukaryotic DNA replication and repair. Therefore, they had to exist prior life began, since DNA replication enzymes and proteins depends on them. They require however also complex proteins and enzymes to be synthesized. Thats a classical chicken/egg problem.
DNA charged with regulating replication
DNA can transport electrical charge over long distances and has the potential to act as a signaling system. The iron-sulfur complex [4Fe4S] found in some proteins is known to be involved in redox reactions. The eukaryotic DNA primase is involved in DNA replication and contains a [4Fe4S] cluster that is required for its RNA primer synthesis activity. O'Brien et al. show that the [4Fe4S] cluster in DNA primase can regulate the protein's DNA binding activity through DNA-mediated charge transfer. This in turn plays a role in primer initiation and length determination.
Orgel summarized his analysis of the proposal by stating,
Self-organizing biochemical cycles
"There is at present no reason to expect that multistep cycles such as the reductive citric acid cycle will self-organize on the surface of FeS/FeS2 or some other mineral."
Recent advances in the Suf Fe-S cluster biogenesis pathway: Beyond the Proteobacteria
Iron-sulfur (Fe-S) cluster metalloproteins play myriad roles in cell function, ranging from amino acid biosynthesis to transcriptional regulation. These diverse functions arise from the multiple types of Fe-S clusters assembled in vivo, ranging from relatively simple [2Fe-2S] clusters, found in some classes of ferredoxin, to complex, mixed-metal clusters, such as the [Mo-7Fe-9S] cluster (or FeMo cofactor) of nitrogenase
Biogenesis of iron-sulfur clusters in mammalian cells: new insights and relevance to human disease
The proteins involved in the biogenesis of Fe-S clusters are conserved from bacteria to humans, and many insights into the process of Fe-S cluster biogenesis have come from studies of model organisms, including bacteria, fungi and plants. Iron-sulfur (Fe-S) clusters are important prosthetic groups with unusual chemical properties that enable the proteins that contain them (Fe-S proteins) to function in pathways ranging from metabolism to DNA repair. They are evolutionarily ancient and are present in essentially all organisms, including Archaea, bacteria, plants and animals. In the anaerobic atmosphere of ancient earth, Fe-S inorganic metal compounds were probably already present in hydrothermal vents
Fe-S cluster biogenesis: the basic pathway
The highly conserved general Fe-S cluster biogenesis pathway has been the subject of intense study in numerous species of bacteria, plants, yeast and mammals since it was first described in bacteria. Many of the general steps of the pathway are common to all kingdoms of life.
The initial stage of Fe-S cluster biogenesis is accomplished by a multimeric protein complex in which a dimer of a cysteine desulfurase called
IscS (Escherichia coli)
Nfs1 (S. cerevisiae)
NFS1 (mammals)
forms a core to which two monomers of a dedicated scaffold protein (IscU in bacteria, Isu1 or Isu2 in yeast and ISCU in humans) bind at either end

A general scheme for biogenesis of Fe-S clusters in mammalian cells.
(A) NFS1 is a cysteine desulfurase that forms a dimer to which monomers of the primary scaffold protein ISCU bind near the top and bottom of the complex.
Mechanism of Sulfur Transfer Across Protein−Protein Interfaces: The Cysteine Desulfurase Model System

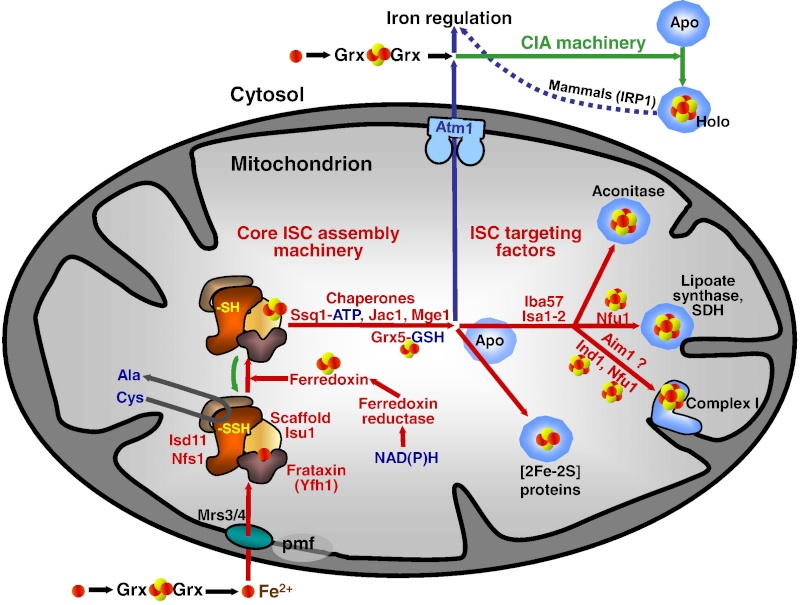
In eukaryotes, ISD11 is an obligate binding partner for NFS1. NFS1 also binds the cofactor pyridoxal phosphate (not shown). Structural and biochemical studies suggest that frataxin forms part of the initial Fe-S cluster biogenesis complex, potentially occupying a pocket between NFS1 and ISCU.NFS1 donates inorganic sulfur, and cysteines from ISCU provide the sulfur ligands that directly bind iron in the nascent Fe-S cluster. A highly reduced protein such as ferredoxin probably provides needed electrons.
(B) Once the Fe-S cluster is assembled, it must be transferred to recipient proteins. Work in bacteria and yeast model systems suggests that a dedicated chaperone–co-chaperone pair of proteins participates in cluster transfer from the primary scaffold, ISCU, to recipient Fe-S proteins. The co-chaperone is known to be HSC20 (a DNAJ protein), whereas the chaperone is an HSP70 homolog that has not yet been clearly identified in mammalian cells. The role of a putative HSC70 protein is proposed here. HSC20 binds ISCU, and the HSC20-ISCU complex probably then binds to its HSC70 partner through two different binding sites: HSC20 contacts the N-terminus of HSC70 and its binding partner, ISCU, binds to the C-terminal substrate-binding domain region of HSC70. The J domain region of HSC20 contains three residues [His (H) Pro (P) and Asp (D); HPD] that activate the ATPase activity of HSC70. Upon activation, a conformational change is proposed to occur in the substrate-binding domain of HSC70 that affects bound ISCU, resulting in extrusion of a peptide containing the residues LPPVK from the ISCU globular protein. The LPPVK peptide then binds to a groove in the substrate-binding domain of HSC70, which consolidates or perhaps further enhances the conformational change in ISCU, which might convert it to a conformation that facilitates donation of its cluster to recipient proteins. In this model, HSC20 helps protect the vulnerable Fe-S cluster bound to ISCU as it dissociates from the multimeric assembly complex, and HSC20 then escorts ISCU to form a trimeric complex with HSC70. The consumption of ATP probably provides a powerful impetus to drive conformational changes of ISCU and the substrate-binding domain of HSC70; these changes might facilitate release of the Fe-S cluster from ISCU. By capturing the energy released by ATP hydrolysis and coupling it to conformational changes, the chaperone–co-chaperone pair help the Fe-S cluster to reach its target proteins. Target proteins could include some direct targets, or proteins such as NFU1, BOLA3, NUBPL or GLRX5 that might function as intermediary scaffolds that then donate Fe-S clusters to specific subsets of recipient proteins.
(C) Mutations in proteins acting at different points in the biogenesis pathway cause diseases with markedly different phenotypes
The ISC assembly systems in bacteria and mitochondria
The experimental study of Fe–S-protein biogenesis was boosted by the identification of the bacterial isc operon27. This discovery not only aided work on bacterial Fe–S-protein assembly, but also influenced the first
attempts to identify biogenesis proteins in eukaryotes. The relationship between bacteria and mitochondria led to the identification and functional characterization of several mitochondrial proteins homologous to the bacterial ISC system . The striking similarities between the bacterial and mitochondrial ISC components and the underlying assembly mechanisms justify a comparative discussion of these related systems (Table 1).

As explained in Box 1, biosynthesis of Fe–S proteins can be separated into two main steps.



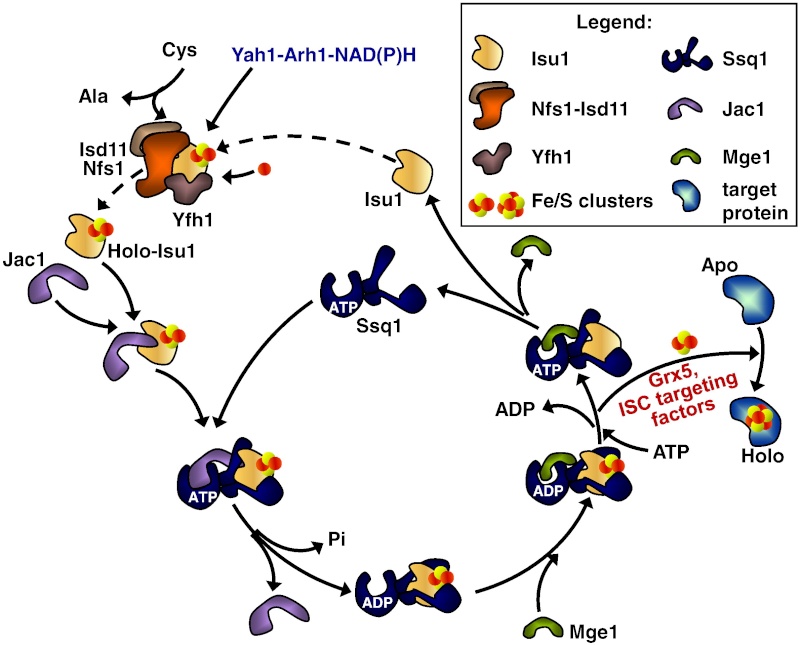
In the ISC systems, an Fe–S cluster is initially and transiently assembled on the scaffold proteins IscU (bacteria) and Isu1 (mitochondria), which contain three conserved Fe–S-cluster-coordinating cysteine residues29–31 (Figs 1 and 2). Then the Fe–S cluster is transferred from Isu1/IscU to recipient apoproteins for incorporation into the Fe–S apoprotein by coordination with specific amino-acid residues . The first reaction, Fe–S-cluster assembly on Isu1/IscU, critically depends on the function of a cysteine desulphurase as a sulphur donor (Box 1). In bacteria, this reaction is performed by IscS, which is highly similar to the founding member of this protein family, NifS, involved in nitrogenase maturation (Fig. 1). The crystal structures of several desulphurases are known and show a dimeric two-domain protein, with one domain harbouring the pyridoxal-phosphate-binding site and a smaller domain containing the active-site cysteine that transiently carries the sulphur released from free cysteine as a persulphide. In mitochondria, the cysteine desulphurase comprises a complex consisting of the IscS-like desulphurase Nfs1 and the 11-kDa protein Isd11 (Fig. 2). Although isolated Nfs1 contains the enzymatic activity as a cysteine desulphurase and releases sulphide from cysteine in vitro, the Nfs1–Isd11 complex is the functional entity for sulphur transfer from Nfs1 to Isu1 in vivo. This reaction is aided by direct interaction between Nfs1 and Isu1 (IscS and IscU in bacteria). On binding of iron to Isu1/IscU, the Fe–S cluster is formed by an unknown mechanism. The iron-binding protein frataxin (Yfh1 in yeast and CyaY in bacteria) is believed to function as an iron donor (Box 1) by undergoing an iron-stimulated interaction with Isu1–Nfs1 . An alternative view recently suggested by in vitro studies is that CyaY functions as an iron-dependent regulator of the biosynthesis reaction by inhibiting IscS43. Fe–S-cluster assembly on Isu1 further depends on electron transfer from the [2Fe–2S] ferredoxin Yah1 (Fdx in bacteria), which receives its electrons from the mitochondrial ferredoxin reductase Arh1 and NADH30 (Fig. 1). It is likely that the electron flow is needed for reduction of the sulphan sulphur (S0) present in cysteine to the sulphide (S2−) present in Fe–S clusters, but this remains to be verified experimentally. An additional electron requirement was suggested for the fusion of two [2Fe–2S] clusters to a [4Fe–4S] cluster by reductive coupling. The second main step of biogenesis formally comprises the release of the Fe–S cluster from Isu1/IscU, cluster transfer to apoproteins and its assembly into the apoprotein. However, these three partial reactions have not been separated experimentally so far. The overall process is specifically assisted by a dedicated chaperone system comprising the Hsp70 ATPase Ssq1 and the DnaJ-like co-chaperone Jac1 (respectively HscA and HscB in bacteria). In mitochondria, the nucleotide exchange factor Mge1 is also required (Fig. 2), whereas in bacteria the related GrpE seems to be dispensable owing to the lability of adenosine diphosphate bound to HscA7. Ssq1/HscA undergoes an ATP-hydrolysis-dependent, highly specific interaction with the LPPVK motif of Isu1/IscU. This complex formation and the involvement of Jac1/HscB is thought to induce a structural change in Isu1/IscU, thereby labilizing Fe–S-cluster binding and, thus, facilitating cluster dissociation and transfer to apoproteins . An ancillary, non-essential role in Fe–S-cluster transfer from Isu1 to apoproteins is performed by the mitochondrial monothiol glutaredoxin Grx5, yet its precise function is unknown. The plant Grx5 proteins were suggested to serve as scaffolds for the formation of [2Fe–2S] clusters. The aforementioned ISC proteins are required for generation of all mitochondrial Fe–S proteins, but some biogenesis components perform a more specific function. The interacting mitochondrial proteins Isa1, Isa2 and Iba57 (Table 1) are specifically involved in the maturation of a subset of Fe–S proteins, that is, members of the aconitase superfamily and radical SAM proteins (Fig. 2). Depletion of these proteins results in corresponding enzyme defects and auxotrophies. Similarly, a deficiency of the Isa-protein-related IscA in bacteria, in conjunction with the homologous SufA (see below; Table 1), affects the assembly of the [4Fe–4S] proteins aconitase and dihydroxy-acid dehydratase, whereas the maturation of some [2Fe–2S] proteins such as ferredoxin is unaltered. The third bacterial member of this protein class, ErpA (Table 1), is essential for growth and involved in the maturation of an Fe–S protein of isoprenoid biosynthesis. Several members of the Isa1/IscA protein family (Table 1) were shown in vitro to bind an Fe–S cluster by means of three conserved cysteine residues in two motifs characterizing these proteins. SufA binds a [2Fe–2S] cluster in vivo that can be transferred to both [2Fe–2S] and [4Fe–4S] proteins in vitro. Together, these observations may support the view that the Isa1/IscA proteins function as alternative scaffolds for a subset of Fe–S proteins (Fig. 1). However, the relative specificity of the Isu1/IscU and Isa1/IscA scaffolds and their functional cooperation will require further scrutiny in vivo to test the physiological relevance of this proposal, particularly because IscA was also shown to bind mononuclear iron4. The mitochondrial P-loop NTPase Ind1 is important for the assembly of respiratory complex I (Fig. 2). On the basis of its homology with the cytosolic scaffold-protein complex Cfd1–Nbp35 ( Table 1), it was proposed that Ind1 serves as a specific scaffold or transfer protein for the assembly of the eight Fe–S clusters into complex I. Consistent with this idea, Ind1 was shown to assemble a labile Fe–S cluster that can be passed on to apoproteins in vitro.
The SUF machinery in bacteria and plastids
Deletion of the isc operon from E. coli is not associated with a major phenotype. Cell viability is affected only when the SUF biogenesis system is simultaneously inactivated. The suf genes are organized in an operon that is induced under iron-limiting and oxidative-stress conditions (Table 1). Gene expression from the isc and suf operons is coordinately regulated by the Fe–S proteins IscR and SufR, which function as transcriptional repressors of their respective operons. During iron deficiency or oxidative stress, the apo form of IscR additionally activates the suf operon. Thereby, both proteins link the efficiency of Fe–S-protein maturation to the extent of gene expression of the two operons. Components of the SUF machinery are found in a variety of prokaryotes, including Archaea and photosynthetic bacteria. The various SUF components fulfil some of the biosynthetic conditions of Fe–S-protein biogenesis (Box 1). A complex of SufS and SufE serves as the cysteine desulphurase (Fig. 1), in which SufS acts similarly to bacterial IscS or NifS and mitochondrial Nfs1–Isd11, but functions mechanistically distinctly. SufE stimulates SufS activity more than tenfold and allows the cysteine-bound persulphide intermediate on SufS to be transferred to a conserved cysteine residue on SufE, from where it is passed on to scaffold proteins. Unexpectedly, SufE has a structure similar to the IscU-type scaffold proteins, but it is not known to function as one. A specific iron donor and an electron requirement (Box 1) in the SUF system are not yet known, but corresponding steps are probably also involved in this pathway. Several SUF proteins may provide a scaffold function for de novo Fe–S-cluster synthesis, but their relative importance and specificity remain to be clarified (Fig. 1). SufA was discussed above as a functional IscA homologue. SufB contains several conserved cysteine residues that can assemble an Fe–S cluster. SufC is an ATPase that is stimulated 100-fold by complex formation with SufB–SufD. Hence, SufC is a likely candidate for a transfer protein facilitating Fe–Scluster delivery from SufB to target proteins (Box 1). Some bacteria contain an IscU-related protein termed SufU that may or may not be encoded in the suf operon. Notably, SufU differs from Isu1/IscU in that it lacks the HscA binding sequence LPPVK of IscU. SUF proteins are also present in plastids, reiterating that this biosynthetic system seems to be less sensitive to high oxygen concentrations. The functionality of plastid SufS, SufE and SufA has been confirmed by in vitro experiments or bacterial complementation studies, but direct experimental evidence for their biogenesis function in planta is usually more difficult to achieve. It should be mentioned in this context that in plastids the SUF proteins may not be the only proteins to support Fe–S protein biogenesis. An important role, possibly as scaffold proteins, is performed by NFU1, NFU2 and NFU3 (also known as Cnfu1, Cnfu2 and Cnfu3), which have homologues in photosynthetic bacteria. NFU proteins show sequence similarity in a 60-residue segment to the C-terminal domain of NifU in bacteria and a similar segment present in Nfu1 in yeast, the function of which is unknown (Fig. 2). In particular, plastid NFU2 has been examined in more detail and shown to function as a scaffold that can assemble a [2Fe–2S] cluster in vitro and transfer it to apoferredoxin. The cnfu2 mutant plants show a dwarf phenotype with faint pale-green leaves and a deficiency in photosystem I and ferredoxins documenting the important role of NFU2 in Fe–S-protein assembly.
Biogenesis of cytosolic and nuclear Fe–S proteins
Fe–S-protein maturation in both the cytosol and the nucleus strictly depends on the function of the mitochondrial ISC assembly machinery (Fig. 3), but the molecular details of this dependence remain to
be defined.

In human cell culture, small amounts of some ISC proteins have been found in the cytosol. A function for the cytosolic human homologue of Isu1 in de novo assembly of cytosolic Fe–S proteins could not be shown, but the protein may play a role in Fe–S-cluster repair after oxidative damage or iron deprivation. Likewise, cytosolic human Nfs1 does not support Fe–S-protein assembly in the cytosol in the absence of mitochondrial Nfs1. The mitochondria-localized ISC assembly machinery is suggested to produce a (still unknown) component (X in Fig. 3) that is exported from the mitochondrial matrix to the cytosol, where it performs an essential function in the maturation process. Because, in particular, Nfs1 is required inside mitochondria to participate in cytosolic and nuclear Fe–S-protein biogenesis in both yeast and human cells, compound X is predicted to be a sulphur-containing moiety. Whether iron is also exported, possibly as part of a preassembled Fe–S cluster, or joins from the cytosol, is currently unknown. The export reaction is accomplished by the ABC transporter Atm1 (ABCB7 in humans) of the mitochondrial inner membrane. Another required component of the export reaction is the sulphydryl oxidase Erv1, located in the intermembrane space. This enzyme has also been shown to catalyse the formation of disulphide bridges in the intermembrane space during Mia40-dependent protein import into the intermembrane space75, and thus performs a dual function. Strikingly, depletion of GSH in yeast shows a similar phenotype as the downregulation of Atm1 or Erv1, that is, defective cytosolic Fe–S-protein biogenesis and increased iron uptake in the cell and mitochondria,whereas the assembly of mitochondrial Fe–S proteins is unaffected. Hence, Atm1, Erv1 and GSH have been described as the ‘ISC export machinery’ (Fig. 3). Maturation of cytosolic and nuclear Fe–S proteins crucially involves the cytosolic Fe–S-protein assembly (CIA) machinery, which comprises five known proteins (Table 1). According to recent in vivo and in vitro studies, this process can be subdivided into two main partial reactions (Fig. 3). First, an Fe–S cluster is transiently assembled on the P-loop NTPases Cfd1 and Nbp35, which form a heterotetrameric complex and serve as a scaffold (Box 1). As mentioned above, this step essentially requires the mitochondrial ISC machineries. From Cfd1– Nbp35, the Fe–S cluster is transferred to apoproteins, a step that requires the CIA proteins Nar1 and Cia1. Cfd1 and Nbp35 take part in the maturation of Nar1 by assisting the assembly of two Fe–S clusters on this irononly hydrogenase-like protein (Fig. 3). Thus, Nar1 is both a target and a component of the CIA machinery, creating a ‘chicken-and-egg’ situation for its maturation process. Nar1 holoprotein assists Fe–S-cluster transfer to target apoproteins by interacting with Cia1, a WD40 repeat protein that serves as a docking platform for binding Nar1 (ref. 79). Recently, another CIA component, termed Dre2, has been identified but its precise molecular function is currently unknown80. The protein coordinates Fe–S clusters, and is probably both a target and a component of the CIA machinery, similar to Nar1. A crucial function of the human homologues of Nar1 and Nbp35 in cytosolic Fe–S-protein biogenesis has been experimentally verified in cultured cells using RNA-interference technology to deplete these proteins to critical level..
Essential enzymes and proteins in FE-S cluster biosynthesis:
Cysteine desulfurase
Ferritin
IscU, CyaY, IscS
Metals in Cells , page 815
1. Evolution of the ferritin family in vertebrates
2. NATURE|Vol 460|13 August 2009|doi:10.1038/nature08301
Sulfur: It’s more important than you might think. We find Sulfur/Iron co-factors throughout life’s chemistry; they may be older than heme or chlorophyll molecules. The possible reasons for this so-called proliferation is the simplicity of the sulfur/iron co-factor’s structure and diversity. We find this co-factor in human biology, plant biology, and insect and bacteria biology. 9
The sulfur cycle is the collection of processes by which sulfur moves to and from minerals (including the waterways)[clarification needed] and living systems. Such biogeochemical cycles are important in geology because they affect many minerals. Biogeochemical cycles are also important for life because sulfur is an essential element, being a constituent of many proteins and cofactors. 2
Iron-sulfur [Fe-S] clusters are ubiquitous, ancient prosthetic groups that are required to sustain fundamental life processes. 6 Iron-sulfur (Fe-S) clusters are required for critical biochemical pathways, including respiration, photosynthesis, and nitrogen fixation. Assembly of these iron cofactors is a carefully controlled process in cells to avoid toxicity from free iron and sulfide.Multiple Fe-S cluster assembly pathways are present in bacteria to carry out basal cluster assembly, stress-responsive cluster assembly, and enzyme-specific cluster assembly.

a | Rhombic iron–sulphur ([2Fe–2S]) clusters are common and are found in many reducing proteins, such as ferredoxins and glutaredoxins101.
b | The ability of two rhombic [2Fe–2S] clusters to coalesce to form a cubane [4Fe–4S] cluster has been documented in vitro26 and in vivo27.
c | The versatile binding characteristics of sulphur are exemplified by its ability to bridge two metal (iron) sites in rhombic [2Fe–2S] clusters, three metal sites in cubane [4Fe–4S] clusters and up to six metal sites for the central sulphur of the complex P-cluster of nitrogenase. The two iron atoms in the top plane of the cubane Fe–S cluster share a blended orbital, in which a single electron is delocalized such that each iron atom has a functional charge of 2.5+, instead of one iron having a charge of 3+ while the other monopolizes a single electron to reduce its charge to 2+. Similar delocalization of an electron is present in the bottom plane (not shown). Delocalization of the added electron between paired iron atoms in a cubane cluster is energetically very favourable because the Fe–S cluster does not need to substantially reorganize its components and ligands to share the electron.
Part c from Beinert, H., Holm, R. H. and Munck, E. Iron–sulfur clusters: nature's modular, multipurpose structures. Science 277, 653–659 (1997). Reprinted with permission from AAAS.
Iron-sulfur (FeS) centers are essential protein cofactors in all forms of life. In particular, FeS centers function as enzyme cofactors in catalysis and electron transfer. Moreover, they are indispensable for the biosynthesis of complex metal centers such as the iron-molybdenum cofactor (FeMoco) of nitrogenase, the molybdenum cofactor of various molybdoenzymes as well as the active sites of [FeFe]- and [Fe]-hydrogenases. In spite of recent fundamental breakthroughs in metalloenzyme research, it has become evident that studies on single enzymes need to be transformed into the broader context of a living cell where biosynthesis, function, and assembly/disassembly of these fascinating metal cofactors are coupled in a dynamic fashion. Various biosynthetic pathways were found to be tightly interconnected through complex crosstalk mechanisms that crucially depend on the bio-availability of the metal ions iron, molybdenum, tungsten, nickel, copper, and zinc. These metals are essential constituents for nitrogenase, hydrogenase and selected molybdo-/tungstoenzymes. Novel methodological developments shall allow for a detailed investigation of the biosynthesis and catalytic function of FeS-dependent enzymes in a cellular context, thus, opening up a new era in metalloenzyme studies. Moreover, cellular studies are a prerequisite for obtaining a comprehensive view on the involvement of metalloenzymes in metal-related human diseases. Understanding the crosstalk of metal ions on a cellular basis requires multidisciplinary and cooperative approaches that span the entire range from cell and molecular biology, biochemistry, inorganic chemistry, spectroscopy, and structural biology to theory. 11
Fe/S proteins in eukaryotes have been localized in mitochondria, cytosol and nucleus where they are involved in rather diverse functions such as the TCA cycle, amino acid biosynthesis, bacterial and mitochondrial respiration, co-factor biosynthesis, ribosome assembly, regulation of protein translation, DNA replication and DNA repair
How and why was this control achieved BEFORE life began ? 10
Many of the general steps of the pathway are common to all kingdoms of life, but it seems that the situation is more complicated in eukaryotes, in which Fe-S proteins are functional and necessary in multiple subcellular compartments, including mitochondria, plastids, cytosol and nucleus. 7
Iron-sulphur clusters are present in more than 200 different types of enzymes or proteins and constitute one of the most ancient, ubiquitous and structurally diverse classes of biological prosthetic groups. Hence the process of iron-sulphur biosynthesis is essential to almost all forms of life and is remarkably conserved in prokaryotic and eukaryotic organisms. Three distinct types of iron-sulphur cluster assembly machinery have been established in bacteria, termed the NIF, ISC and SUF systems 8
In each case the overall mechanism involves cysteine desulphurase-mediated assembly of transient clusters on scaffold proteins and subsequent transfer of preformed clusters to apo proteins. A molecular level understanding of the complex processes of iron-sulphur cluster assembly and transfer is now beginning to emerge from the combination of in vivo and in vitro approaches. The biosynthetic machineries for Fe-S cluster biogenesis are widely conserved in all three kingdoms of life.
The NIF system is generally specific for the maturation of Fe-S proteins in nitrogen fixing organisms such as Azotobacter vinelandii (Av). The ISC system is the primary system for general Fe-S cluster biosynthesis in bacteria such as Escherichia coli and Av . Moreover, along with a few additional components, the ISC system constitutes the eukaryotic mitochondrial machinery for Fe-S cluster biogenesis
How and why did the essential assembly pathways emerge without replication and without evolution, if there would have only been use for the clusters , once duly embedded and inbuilt in the various proteins, essential for life?
The prevalence of these proteins on the metabolic pathways of most organisms leads some scientists to theorize that iron–sulfur compounds had a significant role in the origin of life in the iron–sulfur world theory. 3
The iron–sulfur world hypothesis is a set of proposals for the origin of life and the early evolution of life advanced in a series of articles between 1988 and 1992 by Günter Wächtershäuser, a Munich patent lawyer with a degree in chemistry, who had been encouraged and supported by philosopher Karl R. Popper to publish his ideas. The hypothesis proposes that early life may have formed on the surface of iron sulfide minerals, hence the name.[1][2][3][4][5] It was developed by retrodiction from extant biochemistry in conjunction with chemical experiments. 4
Methionine, cysteine, homocysteine, and taurine are the 4 common sulfur-containing amino acids, but only the first 2 are incorporated into proteins. 5
NIF system FeS cluster assembly, NifU, C-terminal (IPR001075) 10
Iron-sulphur (FeS) clusters are important cofactors for numerous proteins involved in electron transfer, in redox and non-redox catalysis, in gene regulation, and as sensors of oxygen and iron. These functions depend on the various FeS cluster prosthetic groups, the most common being [2Fe-2S] and [4Fe-4S] [PMID: 16221578]. FeS cluster assembly is a complex process involving the mobilisation of Fe and S atoms from storage sources, their assembly into [Fe-S] form, their transport to specific cellular locations, and their transfer to recipient apoproteins. So far, three FeS assembly machineries have been identified, which are capable of synthesising all types of [Fe-S] clusters:
ISC (iron-sulphur cluster),
SUF (sulphur assimilation), and
NIF (nitrogen fixation) systems.
The ISC system is conserved in eubacteria and eukaryotes (mitochondria), and has broad specificity, targeting general FeS proteins [PMID: 16211402, PMID: 16843540]. It is encoded by the isc operon (iscRSUA-hscBA-fdx-iscX). IscS is a cysteine desulphurase, which obtains S from cysteine (converting it to alanine) and serves as a S donor for FeS cluster assembly. IscU and IscA act as scaffolds to accept S and Fe atoms, assembling clusters and transfering them to recipient apoproteins. HscA is a molecular chaperone and HscB is a co-chaperone. Fdx is a [2Fe-2S]-type ferredoxin. IscR is a transcription factor that regulates expression of the isc operon. IscX (also known as YfhJ) appears to interact with IscS and may function as an Fe donor during cluster assembly [PMID: 15937904].
The SUF system is an alternative pathway to the ISC system that operates under iron starvation and oxidative stress. It is found in eubacteria, archaea and eukaryotes (plastids). The SUF system is encoded by the suf operon (sufABCDSE), and the six encoded proteins are arranged into two complexes (SufSE and SufBCD) and one protein (SufA). SufS is a pyridoxal-phosphate (PLP) protein displaying cysteine desulphurase activity. SufE acts as a scaffold protein that accepts S from SufS and donates it to SufA [PMID: 17350000]. SufC is an ATPase with an unorthodox ATP-binding cassette (ABC)-like component. No specific functions have been assigned to SufB and SufD. SufA is homologous to IscA [PMID: 15278785], acting as a scaffold protein in which Fe and S atoms are assembled into [FeS] cluster forms, which can then easily be transferred to apoproteins targets.
In the NIF system, NifS and NifU are required for the formation of metalloclusters of nitrogenase in Azotobacter vinelandii, and other organisms, as well as in the maturation of other FeS proteins. Nitrogenase catalyses the fixation of nitrogen. It contains a complex cluster, the FeMo cofactor, which contains molybdenum, Fe and S. NifS is a cysteine desulphurase. NifU binds one Fe atom at its N-terminal, assembling an FeS cluster that is transferred to nitrogenase apoproteins [PMID: 11498000]. Nif proteins involved in the formation of FeS clusters can also be found in organisms that do not fix nitrogen [PMID: 8875867].
This entry represents the C-terminal of NifU and homologous proteins. NifU contains two domains: an N-terminal (IPR002871) and a C-terminal domain [PMID: 8048161]. These domains exist either together or on different polypeptides, both domains being found in organisms that do not fix nitrogen (e.g. yeast), so they have a broader significance in the cell than nitrogen fixation.
Iron-sulfur clusters: Basic building blocks for life 1
Iron-sulfur (Fe/S) clusters belong to the most ancient co-factors of proteins involved in electron transfer, catalysis and regulatory processes (Beinert et al., 1997). The simplest Fe/S clusters are of the [2Fe-2S] and [4Fe-4S] types which contain either ferrous (Fe2+) or ferric (Fe3+) iron and sulfide (S2-) and which are usually integrated into proteins via coordination of the iron ions by cysteine or histidine residues (Fig. 1A). While (bio)chemists have worked out reconstitution procedures to assemble Fe/S clusters into apoproteins in vitro, cell biological and genetic studies over the past decade have provided ample evidence that the maturation of Fe/S proteins in living cells is a catalyzed rather than spontaneous process.
Despite the chemical simplicity of Fe/S clusters their biosynthesis is rather complex requiring numerous components. Pioneering studies in bacteria have identified three different biosynthesis machineries;the
NIF system for specific maturation of nitrogenase in azototrophic bacteria, the
ISC assembly and the
SUF systems for generation of house-keeping Fe/S proteins under normal and oxidative-stress conditions, respectively (Fontecave et al., 2005; Johnson et al., 2005).
The latter two machineries were inherited by eukaryotes which contain components homologous to those of the bacterial ISC assembly system inside mitochondria (see below; (Lill and Kispal, 2000)) and SUF components in plastids (Balk and Lobreaux, 2005).
‘ISC assembly machinery’.
Fe/S proteins in eukaryotes have been localized in mitochondria, cytosol and nucleus where they are involved in rather diverse functions such as the TCA cycle, amino acid biosynthesis, bacterial and mitochondrial respiration, co-factor biosynthesis, ribosome assembly, regulation of protein translation, DNA replication and DNA repair (Fig. 1B).

The yeast Saccharomyces cerevisiae has served as an excellent model organism to unravel the complex biosynthesis pathways, but recent investigations in human cell culture and transgenic mice have demonstrated that the process is highly conserved in eukaryotes from yeast to man. Since almost a decade my group is dedicated to the identification of the components and mechanisms underlying Fe/S protein biogenesis in eukaryotes using yeast and human cell culture as our major experimental systems (Lill et al., 2006; Lill and Mühlenhoff, 2006). This overview briefly summarizes the principles of how eukaryotic cells generate their Fe/S proteins in the different compartments.
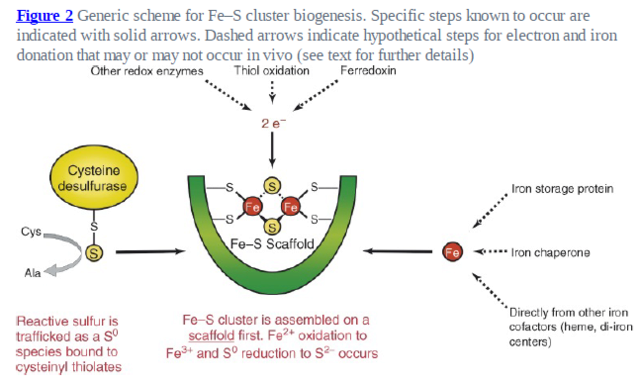
It has been noted early on in the studies of eukaryotic Fe/S protein biogenesis that mitochondria perform a central role as they are required for biogenesis of all cellular Fe/S proteins (Kispal et al., 1999; Schilke et al., 1999). As noted above they harbor the so-called ‘ISC assembly machinery’. To date 15 proteins are known to assist this complex biosynthetic process which can be sub-divided experimentally into two major steps (Fig. 2; (Mühlenhoff et al., 2003)). First, an Fe/S cluster is assembled de novo on the scaffold protein Isu1 which serves as a transient assembly and binding platform. Then, the Fe/S cluster is transferred from Isu1 to recipient apoproteins for incorporation into the Fe/S holoprotein by coordination with specific amino acid residues. Both partial reactions need the assistance of specific ISC assembly components. Only the most important factors will be addressed here. Fe/S cluster assembly on Isu1 critically depends on the function of the cysteine desulfurase complex comprised of Nfs1 and Isd11 (Fig. 2; (Adam et al., 2006; Wiedemann et al., 2006)). Even though Nfs1 contains the enzymatic activity as a cysteine desulfurase and releases sulfur from cysteine to form alanine and a Nfs1-bound persulfide, the Nfs1-Isd11 complex is the functional entity for sulfur transfer from Nfs1 to Isu1 in vivo. This reaction is facilitated by direct interaction of Nfs1 and Isu1. Upon binding of iron to Isu1 the Fe/S cluster is formed by a still unknown biochemical mechanism. Yfh1 (also termed frataxin; Table 1) functions as an iron donor by undergoing an iron-stimulated interaction with Isu1-Nfs1. Iron is imported into the mitochondrial matrix in its reduced form (Fe2+).
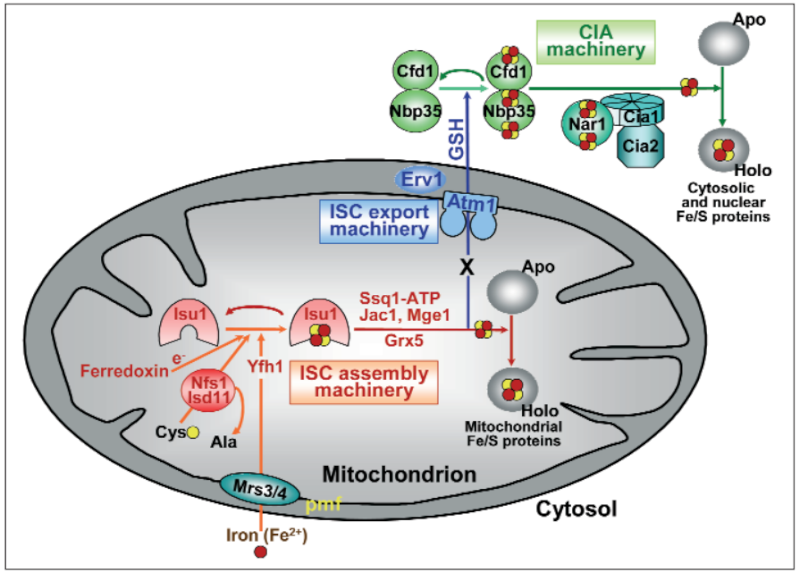
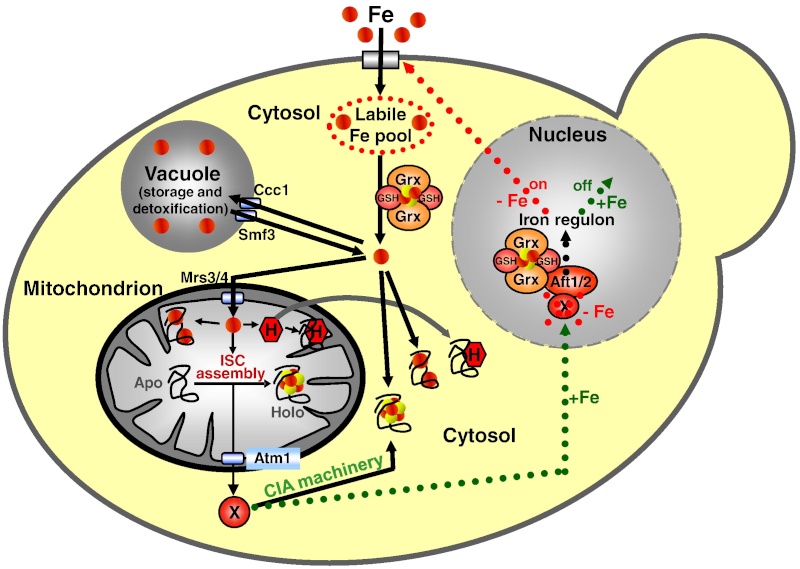
Figure 2: A model for Fe/S protein biogenesis in eukaryotes. Eukaryotic Fe/S protein biogenesis involves the crucial function of mitochondria. The organelles import iron in ferrous (Fe2+, red circle) form from the cytosol in a membrane potential-dependent fashion (pmf). Import is facilitated by the inner membrane carriers Mrs3 and Mrs4. Maturation of mitochondrial Fe/S holoproteins (Holo) involves two major steps. The synthesis of a transiently bound Fe/S cluster (red and yellow circles) on the scaffold protein Isu1 (and Isu2 in yeast) is supported by the early components of the mitochondrial ISC assembly machinery (orange arrows). These proteins include the cysteine desulfurase complex Nfs1-Isd11 which serves as the sulfur (yellow circle) donor for cluster synthesis, the iron binding protein Yfh1 (frataxin) as the iron donor, and the ferredoxin Yah1 which provides electrons (e- ) for sulfur reduction. The release of the Fe/S cluster from Isu1, and its transfer and incorporation into recipient apoproteins (Apo) is facilitated by late components of the ISC assembly machinery (red arrows) including the ATP-dependent Hsp70 chaperone Ssq1, the DnaJ-like cochaperone Jac1, the nucleotide exchange factor Mge1, and the monothiol glutaredoxin Grx5. Extra-mitochondrial Fe/S protein biogenesis requires, in addition to the ISC assembly machinery, components of the mitochondrial ISC export machinery. The ABC transporter Atm1 of the inner membrane exports an unknown compound (X) to the cytosol for use in Fe/S protein assembly (blue arrow), and is assisted by the tripeptide glutathione (GSH) and intermembrane space sulfhydryl oxidase Erv1 which introduces disulfide bridges into substrates. In the cytosol, the CIA machinery catalyzes Fe/S protein maturation in two major steps. First, Fe/S clusters are assembled on the P-loop NTPase complex Cfd1-Nbp35 (light green arrow). The Fe/S clusters are bound to Cfd1-Nbp35 in a labile fashion, and by assistance of Nar1, the WD40 repeat protein Cia1 and Cia2 can be transferred to cytosolic and nuclear apoproteins (dark green arrows).
‘ISC assembly machinery’.
An Fe/S cluster is assembled de novo on the scaffold protein Isu1 . Following protein complexes are required:
1. Cysteine desulfurase complex comprised of Nfs1 and Isd11 Fe/S cluster assembly on Isu1 critically depends on the function of it
2. Yfh1 (also termed frataxin) functions as an iron donor by undergoing an iron-stimulated interaction with Isu1-Nfs1.
3. Proteins Mrs3 and Mrs4 Iron is imported into the mitochondrial matrix in its reduced form (Fe2+).This step requires a membrane potential and requires proteins Mrs3 and Mrs4
4. [2Fe-2S] ferredoxin Yah1 Fe/S cluster assembly on Isu1 further depends on the electron transfer from the [2Fe-2S] ferredoxin Yah1
5. Specific amino acid ligands assembly into the apoprotein by coordination with the specific amino acid ligands.
6. Ssq1
7. Jac1
8. Mge1 its transfer to apoproteins and its assembly into the apoprotein by coordination with the specific amino acid ligands. This step is specifically assisted by a dedicated chaperone system comprised of the Hsp70 family member Ssq1, the DnaJ-like co-chaperone Jac1 and the nucleotide exchange factor Mge1
9. Monothiol glutaredoxin Grx5 Another important function in this partial reaction is performed by the mitochondrial monothiol glutaredoxin Grx5, yet its precise role is unknown hitherto.
10.ISC assembly machinery Apparently, the function of this machinery is critical for the ability of the cell to generate extra-mitochondrial Fe/S proteins,
11. ABC transporter Atm1 The export reaction is accomplished by the ABC transporter Atm1
12. Sulfhydryl oxidase Erv1
13. Glutathione (GSH) The sulfhydryl oxidase Erv1 of the intermembrane space and glutathione (GSH) are required.
14. Cytosolic iron-sulfur protein assembly (CIA) system Maturation of the cytosolic and nuclear Fe/S proteins is catalyzed by the cytosolic iron-sulfur protein assembly (CIA) system comprised of five known proteins
15. Ploop NTPases Cfd1 and Nbp35 An Fe/S cluster is transiently assembled on the Ploop NTPases Cfd1 and Nbp35 which serve as a scaffold.
16. Mitochondria. Ploop NTPases Cfd1 and Nbp35 which serve as a scaffold. This step essentially requires mitochondria.
17. CIA proteins Nar1, Cia1 and Cia2 Cfd1 and Nbp35 involved in activation of CIA protein Nar1 by assembly of two Fe/S clusters on this iron-only hydrogenase-like protein
Essential Fe/S proteins:
1. Essential cytosolic-nuclear Fe/S protein is Rli1 a component involved in ribosome assembly and export from the nucleus
2. Two other essential (nuclear) Fe/S proteins with a function in nucleotide excision repair (Rad3) and RNA primer synthesis for DNA replication (Pri2)
1) https://www.uni-marburg.de/fb20/cyto/lill/publications/pdfs/131.Lill.DGZ.Profil.07.pdf
2) https://en.wikipedia.org/wiki/Sulfur_cycle
3) https://en.wikipedia.org/wiki/Iron%E2%80%93sulfur_protein
4) https://en.wikipedia.org/wiki/Iron%E2%80%93sulfur_world_hypothesis
5) http://jn.nutrition.org/content/136/6/1636S.full
6) http://www.ncbi.nlm.nih.gov/pubmed/15952888
7) http://www.ncbi.nlm.nih.gov/pmc/articles/PMC3291637/
8 http://www.ncbi.nlm.nih.gov/pmc/articles/PMC2827815/
9) http://www.decodedscience.org/sulfur-shade-iron-lifes-origins/40661
10) http://www.ebi.ac.uk/interpro/entry/IPR001075
11) http://www.dfg.de/en/research_funding/announcements_proposals/info_wissenschaft_15_45/index.html
12) http://www.uniprot.org/uniprot/P23503
13) http://www.ncbi.nlm.nih.gov/pubmed/25655665
14) http://www.historyoftheuniverse.com/index.php?p=origlife_2.htm
15) Biological Nitrogen fixation , page 59
Last edited by Admin on Tue Mar 27, 2018 6:43 pm; edited 30 times in total


















 , red (O), blue (N), cyan (Mo), pink (unidentified, likely either N or O in the H-cluster and either C, N, or O in FeMo-co).
, red (O), blue (N), cyan (Mo), pink (unidentified, likely either N or O in the H-cluster and either C, N, or O in FeMo-co).