The Origin and Early History of Life
There are a great many scientists with intriguing ideas that explain how life may have originated on earth, but there is very little that we know for sure. New hypotheses are being proposed constantly, and old ones reevaluated. The oldest clear evidence of life—microfossils in ancient rock—are 3.5 billion years old. The origin of life seems to have taken just the right combination of physical events and chemical processes.
Theories about the Origin of Life
If life did not originate at the ocean’s edge under the blanket of a reducing atmosphere, where did it originate?
Under frozen oceans. One hypothesis proposes that life originated under a frozen ocean, not unlike the one that covers Jupiter’s moon Europa today. All evidence suggests, however, that the early earth was quite warm and frozen oceans quite unlikely.
Deep in the earth’s crust. Another hypothesis is that life originated deep in the earth’s crust. In 1988 Gunter Wachtershauser proposed that life might have formed as a by-product of volcanic activity, with iron and nickel sulfide minerals acting as chemical catalysts to recombine gases spewing from eruptions into the building blocks of life. In later work he and coworkers were able to use this unusual chemistry to build precursors for amino acids (although they did not actually succeed in making amino acids), and to link amino acids together to form peptides. Critics of this hypothesis point out that the concentration of chemicals used in their experiments greatly exceed what is found in nature
Within clay. Other researchers have proposed the unusual hypothesis that life is the result of silicate surface chemistry. The surface of clays have positive charges to attract organic molecules, and exclude water, providing a potential catalytic surface on which life’s early chemistry might have occurred. While interesting conceptually, there is little evidence that this sort of process could actually occur.
At deep-sea vents. Becoming more popular is the hypothesis that life originated at deep-sea hydrothermal vents, with the necessary prebiotic molecules being synthesized on metal sulfides in the vents. The positive charge of the sulfides would have acted as a magnet for negatively charged organic molecules. In part, the current popularity of this hypothesis comes from the new science of genomics, which suggests that the ancestors of today’s prokaryotes are most closely related to the bacteria that live on the deep-sea vents.
Origins of Life, Hugh Ross:
naturalistic models share some “controlling” central features, including natural pathways for:
• synthesis (combining) of prebiotic (pre-life) molecules
• concentration of prebiotic molecules
• formation of life’s building blocks
• assembly of building block molecules to form complex biomolecules
• development of self-replication
• emergence of metabolism (the physical and chemical processes continuously going on in living organisms and cells)
• aggregation of biomolecules to form protocells (primitive cells)
• evolution of protocells into true cells
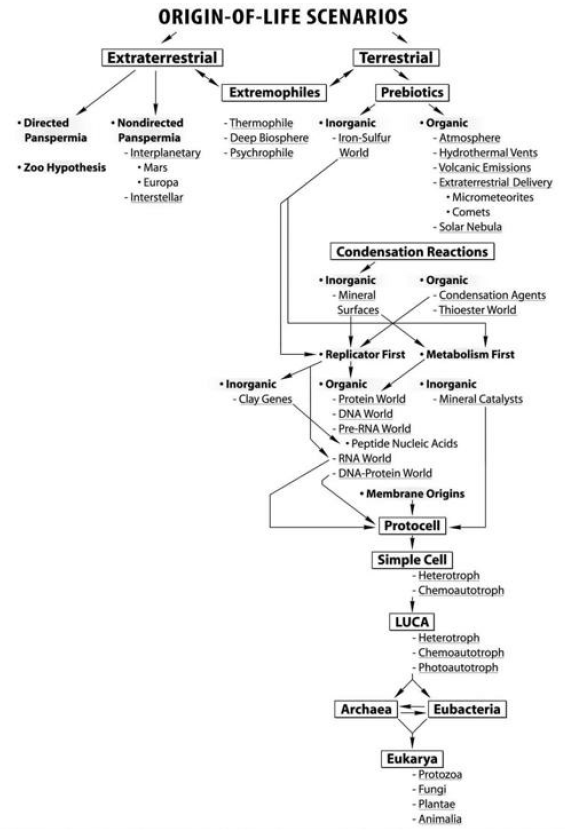
Origin of Life
1. The principle of common ancestry states that all life on Earth can be traced back to a single ancestor, the last universal common ancestor (LUCA).
2. All living things:
a. Acquire energy through metabolism.
b. Respond and interact with their environment.
c. Self-replicate.
d. Are subject to the forces of natural slection that drive adaptation to the environment.
3. The molecules of living things are called biomolecules and are organic.
a. The four stages of the origin of life are:
i. Organic monomers
ii. Organic polymers
iii. Protocells
iv. Living cells
A. Stage 1. Evolution of Monomers
1. There are three hypotheses that explain how organic monomers could have evolved.
2. Hypothesis one: monomers came from reactions in the atmosphere.
a. Oparin/Haldane independently suggested organic molecules could be formed in the presence of outside energy sources using atmospheric gases.
i. Their hypothesis is sometimes called the “primordial soup” hypothesis.
ii. The reducing atmosphere of early Earth could have driven abiotic synthesis of organic monomers from inorganic molecules.
b. Experiments performed by Miller and Urey (1953) showed experimentally that these gases (methane, ammonia, hydrogen, water) reacted with one another to produce small organic molecules (amino acids, organic acids).
3. Hypothesis two: monomers came from reactions in ocean thermal vents.
a. Günter Wächtershäuser came up with the iron-sulfur world hypothesis in the late 1980s.
b. The ocean’s thermal vents have iron and nickel sulfide minerals present, and the vents emit gases such as carbon monoxide, ammonia, and hydrogen sulfide.
c. The iron and nickel sulfide act as catalysts that drive the chemical evolution from inorganic to organic molecules.
4. Hypothesis three: monomers came from outer space.
a. Comets and meteorites, perhaps carrying organic chemicals, have pelted the Earth throughout history.
b. A meteorite from Mars (ALH84001) that landed on Earth 13,000 years ago, may have fossilized bacteria.
B. Stage 2. Evolution of Polymers
1. Hypothesis one: iron-sulfur world hypothesis
a. Wächtershäuse and Huber formed peptides using iron-nickel sulfides under vent-like conditions.
b. Such minerals have a charged surface that attracts amino acids and provides electrons so they bond together.
2. Hypothesis two: protein-first hypothesis
a. Sidney Fox demonstrated amino acids polymerize abiotically if exposed to dry heat.
b. Amino acids could have collected in shallow puddles along the rocky shore and then formed proteinoids (i.e., small polypeptides that have some catalytic properties) from the heat of the sun.
c. When proteinoids are placed in water, they form cell-like microspheres composed of protein.
d. This assumes DNA genes came after protein enzymes; DNA replication needs protein enzymes.
3. Hypothesis three: RNA-first hypothesis
a. Only the macromolecule RNA was needed at the beginning to lead to the first cell.
b. Thomas Cech and Sidney Altman discovered that RNA can be both a substrate and an enzyme.
c. RNA would carry out processes of life associated with DNA (in genes) and protein enzymes.
d. Supporters of this hypothesis label 4 BYA an “RNA world.”
C. Stage 3. Evolution of Protocells
1. Before the first true cell arose, there would have been a protocell or protobiont.
2. A protocell would have a lipid-protein membrane and carry on energy metabolism.
3. Sidney Fox showed that if lipids are made available to microspheres, lipids become associated with microspheres producing a lipid-protein membrane.
4. Oparin demonstrated a protocell could have developed from coacervate droplets.
a. Coacervate droplets are complex spherical units that spontaneously form when concentrated mixtures of macromolecules are held in the right temperature, ionic composition, and pH.
b. Coacervate droplets absorb and incorporate various substances from the surrounding solution.
5. Aleg Bangham discovered that lipids would naturally organize themselves into double-layered bubbles, known as liposomes.
6. David Deamer and Bangham thought liposomes provided life’s first membranous boundary, and suggested the membrane-first hypothesis.
a. The membrane-first hypothesis states that the first cell had to have a plasma membrane before any of its other parts.
c. In a liquid environment, phospholipid molecules spontaneously form liposomes, spheres surrounded by a layer of phospholipids; this supports the membrane-first hypothesis.
d. A protocell could have contained only RNA to function as both genetic material and enzymes.
7. If a protocell was a heterotrophic fermenter living on the organic molecules in the organic soup that was its environment, this would indicate heterotrophs preceded autotrophs.
a. A heterotroph is an organism that cannot synthesize organic compounds from inorganic substances and therefore must take in preformed organic compounds.
b. An autotroph is an organism that makes organic molecules from inorganic nutrients.
8. If the protocell evolved at hydrothermal vents, it would be chemosynthetic and autotrophs would have preceded heterotrophs.
9. The first protocells may have used preformed ATP, but as supplies dwindled, natural selection would favor cells that could extract energy from carbohydrates to transform ADP to ATP.
10. Since glycolysis is a common metabolic pathway in living things, it evolved early in the history of life.
11. As there was no free O2, it is assumed that protocells carried on a form of fermentation.
12. The first protocells had a limited ability to break down organic molecules; it took millions of years for glycolysis to evolve completely.
D. Stage 4. Evolution of a Self-Replication System
1. In living systems, information flows from DNA → RNA → protein; it is possible that this sequence developed in stages.
2. The RNA-first hypothesis suggests that the first genes and enzymes were RNA molecules.
a. These genes would have directed and carried out protein synthesis.
b. Ribozymes are RNA that acts as enzymes.
c. Some viruses contain RNA genes with a protein enzyme called reverse transcriptase that uses RNA as a template to form DNA; this could have given rise to the first DNA.
3. The protein-first hypothesis contends that proteins or at least polypeptides were the first to arise.
a. Only after the protocell develops complex enzymes could it form nucleic acids from small molecules.
b. Because a nucleic acid is complicated, the chance that it arose on its own is minimal.
c. Therefore, enzymes are needed to guide the synthesis of nucleotides and then nucleic acids.
4. Cairns-Smith suggests that polypeptides and RNA evolved simultaneously.
a. The first true cell would contain RNA genes that replicated because of the presence of proteins; they become associated in clay in such a way that the polypeptides catalyzed RNA formation.
b. This eliminates the chicken-and-egg paradox; both events happen at the same time.
5. Once the protocell was capable of reproduction, it became a true cell and biological evolution began.
a. After DNA formed, the genetic code still had to evolve to store information.
b. Because the current code is subject to fewer errors than other possible codes, and because it minimizes mutations, it likely underwent a natural selection process.
There are a great many scientists with intriguing ideas that explain how life may have originated on earth, but there is very little that we know for sure. New hypotheses are being proposed constantly, and old ones reevaluated. The oldest clear evidence of life—microfossils in ancient rock—are 3.5 billion years old. The origin of life seems to have taken just the right combination of physical events and chemical processes.
Theories about the Origin of Life
If life did not originate at the ocean’s edge under the blanket of a reducing atmosphere, where did it originate?
Under frozen oceans. One hypothesis proposes that life originated under a frozen ocean, not unlike the one that covers Jupiter’s moon Europa today. All evidence suggests, however, that the early earth was quite warm and frozen oceans quite unlikely.
Deep in the earth’s crust. Another hypothesis is that life originated deep in the earth’s crust. In 1988 Gunter Wachtershauser proposed that life might have formed as a by-product of volcanic activity, with iron and nickel sulfide minerals acting as chemical catalysts to recombine gases spewing from eruptions into the building blocks of life. In later work he and coworkers were able to use this unusual chemistry to build precursors for amino acids (although they did not actually succeed in making amino acids), and to link amino acids together to form peptides. Critics of this hypothesis point out that the concentration of chemicals used in their experiments greatly exceed what is found in nature
Within clay. Other researchers have proposed the unusual hypothesis that life is the result of silicate surface chemistry. The surface of clays have positive charges to attract organic molecules, and exclude water, providing a potential catalytic surface on which life’s early chemistry might have occurred. While interesting conceptually, there is little evidence that this sort of process could actually occur.
At deep-sea vents. Becoming more popular is the hypothesis that life originated at deep-sea hydrothermal vents, with the necessary prebiotic molecules being synthesized on metal sulfides in the vents. The positive charge of the sulfides would have acted as a magnet for negatively charged organic molecules. In part, the current popularity of this hypothesis comes from the new science of genomics, which suggests that the ancestors of today’s prokaryotes are most closely related to the bacteria that live on the deep-sea vents.
Origins of Life, Hugh Ross:
naturalistic models share some “controlling” central features, including natural pathways for:
• synthesis (combining) of prebiotic (pre-life) molecules
• concentration of prebiotic molecules
• formation of life’s building blocks
• assembly of building block molecules to form complex biomolecules
• development of self-replication
• emergence of metabolism (the physical and chemical processes continuously going on in living organisms and cells)
• aggregation of biomolecules to form protocells (primitive cells)
• evolution of protocells into true cells

Origin of Life
1. The principle of common ancestry states that all life on Earth can be traced back to a single ancestor, the last universal common ancestor (LUCA).
2. All living things:
a. Acquire energy through metabolism.
b. Respond and interact with their environment.
c. Self-replicate.
d. Are subject to the forces of natural slection that drive adaptation to the environment.
3. The molecules of living things are called biomolecules and are organic.
a. The four stages of the origin of life are:
i. Organic monomers
ii. Organic polymers
iii. Protocells
iv. Living cells
A. Stage 1. Evolution of Monomers
1. There are three hypotheses that explain how organic monomers could have evolved.
2. Hypothesis one: monomers came from reactions in the atmosphere.
a. Oparin/Haldane independently suggested organic molecules could be formed in the presence of outside energy sources using atmospheric gases.
i. Their hypothesis is sometimes called the “primordial soup” hypothesis.
ii. The reducing atmosphere of early Earth could have driven abiotic synthesis of organic monomers from inorganic molecules.
b. Experiments performed by Miller and Urey (1953) showed experimentally that these gases (methane, ammonia, hydrogen, water) reacted with one another to produce small organic molecules (amino acids, organic acids).
3. Hypothesis two: monomers came from reactions in ocean thermal vents.
a. Günter Wächtershäuser came up with the iron-sulfur world hypothesis in the late 1980s.
b. The ocean’s thermal vents have iron and nickel sulfide minerals present, and the vents emit gases such as carbon monoxide, ammonia, and hydrogen sulfide.
c. The iron and nickel sulfide act as catalysts that drive the chemical evolution from inorganic to organic molecules.
4. Hypothesis three: monomers came from outer space.
a. Comets and meteorites, perhaps carrying organic chemicals, have pelted the Earth throughout history.
b. A meteorite from Mars (ALH84001) that landed on Earth 13,000 years ago, may have fossilized bacteria.
B. Stage 2. Evolution of Polymers
1. Hypothesis one: iron-sulfur world hypothesis
a. Wächtershäuse and Huber formed peptides using iron-nickel sulfides under vent-like conditions.
b. Such minerals have a charged surface that attracts amino acids and provides electrons so they bond together.
2. Hypothesis two: protein-first hypothesis
a. Sidney Fox demonstrated amino acids polymerize abiotically if exposed to dry heat.
b. Amino acids could have collected in shallow puddles along the rocky shore and then formed proteinoids (i.e., small polypeptides that have some catalytic properties) from the heat of the sun.
c. When proteinoids are placed in water, they form cell-like microspheres composed of protein.
d. This assumes DNA genes came after protein enzymes; DNA replication needs protein enzymes.
3. Hypothesis three: RNA-first hypothesis
a. Only the macromolecule RNA was needed at the beginning to lead to the first cell.
b. Thomas Cech and Sidney Altman discovered that RNA can be both a substrate and an enzyme.
c. RNA would carry out processes of life associated with DNA (in genes) and protein enzymes.
d. Supporters of this hypothesis label 4 BYA an “RNA world.”
C. Stage 3. Evolution of Protocells
1. Before the first true cell arose, there would have been a protocell or protobiont.
2. A protocell would have a lipid-protein membrane and carry on energy metabolism.
3. Sidney Fox showed that if lipids are made available to microspheres, lipids become associated with microspheres producing a lipid-protein membrane.
4. Oparin demonstrated a protocell could have developed from coacervate droplets.
a. Coacervate droplets are complex spherical units that spontaneously form when concentrated mixtures of macromolecules are held in the right temperature, ionic composition, and pH.
b. Coacervate droplets absorb and incorporate various substances from the surrounding solution.
5. Aleg Bangham discovered that lipids would naturally organize themselves into double-layered bubbles, known as liposomes.
6. David Deamer and Bangham thought liposomes provided life’s first membranous boundary, and suggested the membrane-first hypothesis.
a. The membrane-first hypothesis states that the first cell had to have a plasma membrane before any of its other parts.
c. In a liquid environment, phospholipid molecules spontaneously form liposomes, spheres surrounded by a layer of phospholipids; this supports the membrane-first hypothesis.
d. A protocell could have contained only RNA to function as both genetic material and enzymes.
7. If a protocell was a heterotrophic fermenter living on the organic molecules in the organic soup that was its environment, this would indicate heterotrophs preceded autotrophs.
a. A heterotroph is an organism that cannot synthesize organic compounds from inorganic substances and therefore must take in preformed organic compounds.
b. An autotroph is an organism that makes organic molecules from inorganic nutrients.
8. If the protocell evolved at hydrothermal vents, it would be chemosynthetic and autotrophs would have preceded heterotrophs.
9. The first protocells may have used preformed ATP, but as supplies dwindled, natural selection would favor cells that could extract energy from carbohydrates to transform ADP to ATP.
10. Since glycolysis is a common metabolic pathway in living things, it evolved early in the history of life.
11. As there was no free O2, it is assumed that protocells carried on a form of fermentation.
12. The first protocells had a limited ability to break down organic molecules; it took millions of years for glycolysis to evolve completely.
D. Stage 4. Evolution of a Self-Replication System
1. In living systems, information flows from DNA → RNA → protein; it is possible that this sequence developed in stages.
2. The RNA-first hypothesis suggests that the first genes and enzymes were RNA molecules.
a. These genes would have directed and carried out protein synthesis.
b. Ribozymes are RNA that acts as enzymes.
c. Some viruses contain RNA genes with a protein enzyme called reverse transcriptase that uses RNA as a template to form DNA; this could have given rise to the first DNA.
3. The protein-first hypothesis contends that proteins or at least polypeptides were the first to arise.
a. Only after the protocell develops complex enzymes could it form nucleic acids from small molecules.
b. Because a nucleic acid is complicated, the chance that it arose on its own is minimal.
c. Therefore, enzymes are needed to guide the synthesis of nucleotides and then nucleic acids.
4. Cairns-Smith suggests that polypeptides and RNA evolved simultaneously.
a. The first true cell would contain RNA genes that replicated because of the presence of proteins; they become associated in clay in such a way that the polypeptides catalyzed RNA formation.
b. This eliminates the chicken-and-egg paradox; both events happen at the same time.
5. Once the protocell was capable of reproduction, it became a true cell and biological evolution began.
a. After DNA formed, the genetic code still had to evolve to store information.
b. Because the current code is subject to fewer errors than other possible codes, and because it minimizes mutations, it likely underwent a natural selection process.
Last edited by Admin on Sat Aug 29, 2015 7:08 pm; edited 2 times in total














