https://reasonandscience.catsboard.com/t1694-no-evidence-for-the-evolution-of-birds-feathers-and-flight
It's ridiculous to think that birds "evolved" from reptiles. There are many features besides feathers that enable birds to fly. Their brains had to be modified for flight. Their bones had to have the hollow lattice as seen at present, and they had to develop special lungs that enable them to breathe deeply at every stroke of their wings. All of these structures, besides specialized development of the feather itself is necessary for a bird to fly. This coordination of characteristics makes the idea that all these flight features evolved randomly into the bird we see today profoundly absurd.
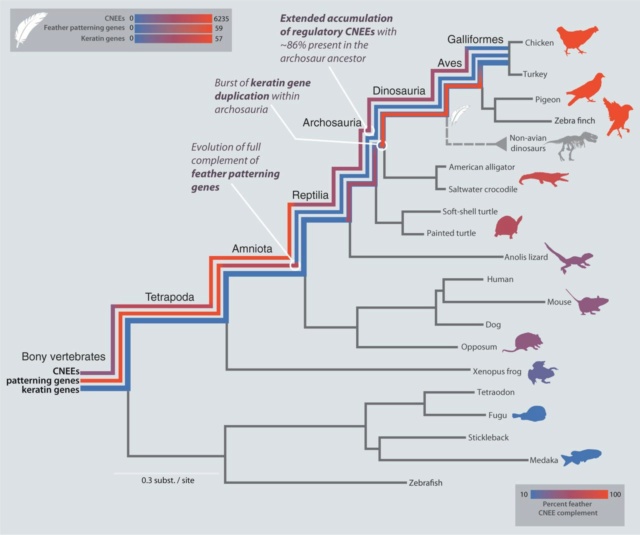
The anatomical placode in reptile scale morphogenesis indicates shared ancestry among skin appendages in amniotes 24 Jun 2016:
The fossil record lacks any evidence of intermediate forms (hence, of homology) between scales and hairs. In addition, hairs in mammals, feathers and feet scales in birds, and scales in reptiles exhibit substantial differences in morphogenesis. Finally, the presence and absence of β-proteins [a family of proteins unrelated to α-keratins ] in skin appendages of sauropsids (birds and reptiles) and in those of synapsids (mammals), respectively, only added to the confusion. All these considerations have, for decades, fostered the debate on the homology, or lack thereof, among these skin appendages and led some authors to conclude that homologous skin appendages do not exist beyond amniote classes (reptiles, mammals, and birds); that is, mammalian hair and avian feather would not have evolved from reptilian overlapping scales.
https://advances.sciencemag.org/content/2/6/e1600708
Take flying birds for example; suppose you aren't one, and you want to become one. You'll need a baker's dozen highly specialized systems, including wings, flight feathers, the specialized system which allows flight feathers to pivot so as to open on upstrokes and close to trap air on downstrokes (like a venetian blind), a specialized light bone structure, specialized flow-through design heart and lungs, specialized tail, specialized general balance parameters etc.
For starters, every one of these things would be antifunctional until the day on which the whole thing came together, so that the chances of evolving any of these things by any process resembling evolution (mutations plus selection) would amount to an infinitessimal, i.e. one divided by some gigantic number.
In probability theory, to compute the probability of two things happening at once, you multiply the probabilities together. That says that the likelihood of all these things ever happening, best case, is ten or twelve such infinitessimals multiplied together, i.e. a tenth or twelth-order infinitessimal. The whole history of the universe isn't long enough for that to happen once.
All of that was the best case. In real life, it's even worse than that. In real life, natural selection could not plausibly select for hoped-for functionality, which is what would be required in order to evolve flight feathers on something which could not fly apriori. In real life, all you'd ever get would some sort of a random walk around some starting point, rather than the unidircetional march towards a future requirement which evolution requires.
And the real killer, i.e. the thing which simply kills evolutionism dead, is the following consideration: In real life, assuming you were to somehow miraculously evolve the first feature you'd need to become a flying bird, then by the time another 10,000 generations rolled around and you evolved the second such feature, the first, having been disfunctional/antifunctional all the while, would have DE-EVOLVED and either disappeared altogether or become vestigial.
Now, it would be miraculous if, given all the above, some new kind of complex creature with new organs and a new basic plan for life had ever evolved ONCE.
Evolutionism, however (the Theory of Evolution) requires that this has happened countless billions of times, i.e. an essentially infinite number of absolutely zero probability events.
I ask you: What could be stupider than that?
http://www.veritas-ucsb.org/library/origins/quotes/macroevolution.html
"Feathers are features unique to birds, and there are no known intermediate structures between reptilian scales and feathers. Notwithstanding speculations on the nature of the elongated scales found on such forms as Longisquama ... as being featherlike structures, there is simply no demonstrable evidence that they in fact are. They are very interesting, highly modified and elongated reptilian scales, and are not incipient feathers."
Feduccia, Alan (1985) "On Why Dinosaurs Lacked Feathers The Beginning of Birds Eichstatt, West Germany: Jura Mus
As one Columbia University biologist put it, “ . . . we lack completely fossils of all intermediate stages between reptilian scales and the most primitive feather.”
http://cryptozoologynews.blogspot.com.br/2011/07/bird-fossils-reveal-lifes-colourful.html
Oregon State University, “Discovery raises new doubts about dinosaur-bird links,” June 9th, 2009, Physorg.com.
http://www.physorg.com/news163760732.html
The origin of birds has always been a major problem for Darwinism, and even today little agreement about the evolution of birds exists. One of the most difficult issues related to bird evolution is the evolution of feathers. Feathers are complex, designed structures required for flight, and are today found only on birds. A literature review on the evolution of bird feathers showed that even though feathers are found back as far as the Cretaceous, including many well-preserved samples in amber, the fossil record reveals a complete absence of evidence for feather evolution.
http://crev.info/2014/02/story-on-evolution-of-birds-glosses-over-details/
The beautiful Illustra documentary Flight: The Genius of Birds gives a much more elegant and satisfying explanation for flight, because it doesn’t gloss over the details, but accounts for all the traits needed for powered flight: efficient one-way lungs, efficient digestive and excretory systems, the beautifully engineered flight muscles that provide a compact center of gravity, the hollow bones, the navigation systems, the sensory components (able not only to see details from the air but to sense the magnetic field), the exquisite design of feathers, and the behaviors that allow birds to take advantage of air currents, including the lift from other birds in formation flight (1/16/14). The integrated systems that allow an eagle to pick a fish out of a lake, a hummingbird (12/05/13) to hover in mid-air sucking its food out of a flower with a specialized nectar-trapping tongue, or a snowy egret with its large, elegant wings to fly between tree limbs without hitting them, are explained by appealing to what we know in our universal experience about complex functional systems. Intelligent design is a known “vera causa” (true cause) that can account for the observations.
Evolution, by contrast, comes up empty looking for a true cause for flight (7/30/13). Never do we see blind, unguided processes leading to complex functional systems with integrated parts contributing to the overall design goal. Intelligent design is, therefore, the best scientific explanation in contrast to the storytelling from the Darwin camp (9/30/13). The Tweety Rex fable looks downright silly by comparison. With its high perhapsimaybecouldness index, its stretchable rates of evolution (a clear ad hoc theory rescue device), and its copious use of magic words (emerged, arose, developed, appeared), it reduces to “birds evolved because they evolved.” If the public were allowed to hear the two explanations side by side, there would be no contest. Darwin’s flightless DODO birds would go running out of the auditorium in shame.
http://able2know.org/topic/232172-1
Consider feathers, which come in more than one form. Down feathers serve for insulation and are not that much different from hair or fur. A proponent of evolution could talk about fur mutating into down feathers and not sound totally stupid. But flight feathers are so totally different from down feathers that you'd need TWO mutations to get to them i.e. one mutation to get from fur to down feathers and then another to get from down feathers to flight feathers.
http://www.birds.cornell.edu/AllAboutBirds/studyingbirdsi/feather_detail.gif
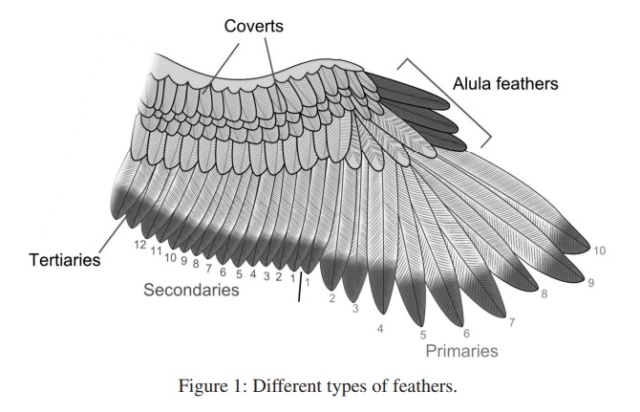
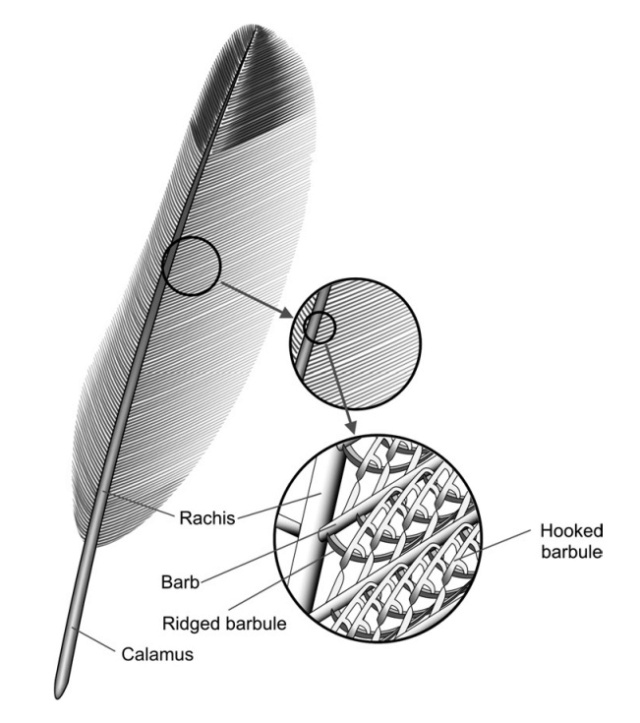
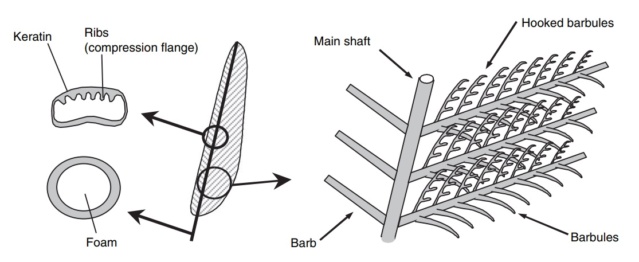
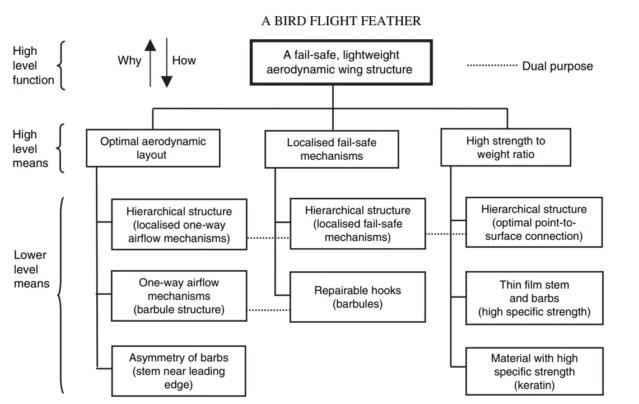
Flight feathers are asymmetric (one side shorter than other) and they pivot so as to open and let air pass through on upstrokes and close again on down-strokes and a the short side is the locking side. Flight feathers involve a complex system of barbules and hooks to create the strength needed to bear weight. Down feathers don't have any of that stuff.
The question is, what kind of a mutation would cause down feathers to mutate into flight feathers ONLY ON THE CREATURE'S ARMS where they will be needed after other mutations turn those arms into wings??
What Is the Evidence for Feathers Before Flight?
Feathers are branched structures consisting of β-keratin—a rigid protein material formed by pleated β sheets—with a hollow central shaft. They are strikingly different from other forms of vertebrate integument such as scales, skin, and hair. Until recently, evolutionary hypotheses envisioned their origin through elongation of broad, flat scales driven by selection for aerial locomotion such as gliding or flapping flight. Over the course of the past two decades, fossil discoveries, especially from northeast China, have revealed that the early precursors of feathers were filament-like rather than expanded scales and that branched pinnate feathers of modern aspect predate the origin of active flight.
The Chinese deposits provide one such unique snapshot, where over a thousand specimens with fine details of soft tissues such as feathers, hair, and skin are preserved in ash-rich lake deposits ranging from the Late Jurassic (∼150 million years ago) to the Early Cretaceous (∼120 million years ago). Fossils from these deposits have revealed that dinosaurs that were inferred from bone characteristics to be closely related to living birds also share more features of feather structure.
Oh, wow, this article is so classic of the evolutionary genre, it’s a virtual gift to creationists. Aside from the obvious evidential conclusion that dinosaur-to-bird evolution is a myth, Clarke used all the evolutionary tricks of the trade we’ve been pointing out in the Darwin lit for 12 years now: the Stuff Happens law, just-so stories, shielding complex changes in words like “novelty” and “innovation,” promissory notes, the coulda-woulda-shoulda habit, embedding evolution in terms like “protofeathers,” the convergence concoction, using passive verbs and subjunctive mood as covers for ignorance, composite explanations, punk eek, incredible stasis, ghost lineages, “more research is needed,” job security for storytellers, glossing over soft tissue in supposedly ancient material, tidbits of Lamarck and Haeckel as needed when gradualism doesn’t work, forcing uncooperative data into prefab scenarios, parading naked Emperor Charlie in public, sacrificing brains at his shrine – everything. Hardly a sentence of this article is devoid of fallacies masquerading as science.
We hope you caught these things before the commentary began. If not, you need Creation-Evolution Headlines as a deprogramming course. Bookmark this site and come for your daily therapy. Since Ken Dial made an off-camera appearance, we like all beginners to get the shock treatment in our commentary from 12/22/03 – the first time (now a decade ago, still cited favorably by Darwinists) the Montana drunkard-on-Darwine presented living partridge family chicks as possible props to an evolutionary tale. Read that color commentary now; know the tricks, and you won’t be fooled again.
The Evolution of Feathers: A Major Problem for Darwinism
http://www.answersingenesis.org/articles/tj/v17/n1/feathers
Even though fossil impressions of feathers are abundant in the fossil record, and much has been written speculating on how scale-to-feather evolution could have occurred, not a shred of fossil or other evidence has ever been found to support the scale-to-feather evolution theory.1,23 In the words of Prum, understanding ‘the evolutionary origin of feathers has been constrained by the lack of any known ancestral feather morphologies or structural antecedents’.41
The evidence supports Klotz’s early conclusion that the ‘origin of feathers is still a real problem’ for Darwinism, and all contemporary theories of feather origin are hypothetical ideas that ‘can only be characterized as judicious speculation’.75 In short, nothing has changed since Regal stated ‘although most textbooks include some sort of speculation on the evolutionary origin of feathers … [a] morass of contradictory theories and muddy thinking … occurs in … much of the literature on this subject’.31
Although much speculation and major disagreements exist on how feathers ‘could have’ evolved, all existing theories are ‘just-so stories’, unsupported by fossil or historical evidence. The profound evolutionary enigma of feathers noted by Darwin76 and Heilmann77 remains, even today. The lack of evidence for feather evolution is not only a major problem for Darwinism, but the design and function of feathers provides evidence for both intelligent design and irreducible complexity. Flight and feathers are indeed a ‘miracle’.78 Feather evolution is related to the question of bird evolution.
Periodically, new bird fossils are found, but most of them have been of little or no use as evidence of bird evolution, and the few claimed examples typically generate much debate. For instance, Feduccia concluded that one recent find, known as Apsaravis, contributes little
‘… to our understanding of avian evolution, and its lack of a clear relationship with any kind of modern bird makes its significance ambiguous. If Apsaravis is not related to any modern ornithurine, how can it tell us anything important about the evolutionary questions raised by [its discoverers] Norell and Clarke?’79
The latest discovery of feathers on the birdlike, turkey-sized ‘theropods’ Caudipteryx and Protarchaeopteryx indicate that they are flightless birds. Much debate exists about this and related finds.80 Some consider these animals to be birdlike dinosaurs, or other dinosaur-like, flightless birds that have lost their full flight plumage (or never developed it). Conclusions on these finds will require much more study, and yet already have produced much debate and controversy.
Much disagreement still exists about Archaeopteryx, a discovery now around 150 years old. Likewise, the place in evolution, if any, of the recent finds may never be settled. Many of these finds are from a province of China, and already one find from this area has proven to be a hoax. Consequently, much more study is necessary to determine the value of these finds. So far, none of these finds challenges the conclusions presented in this paper, and early study of these finds has strongly supported the findings reviewed here.
In conclusion, we agree with Brush: ‘Uncountable numbers of words have been written in attempts to … reconstruct the primitive feather and explain why feathers evolved’.56 So far, all of these attempts have not only failed, but also have led to the conclusion that how feathers ‘arose initially, presumably from reptilian scales, defies analysis’.81
http://www.evolutionnews.org/2011/01/peer-reviewed_pro-intelligent042261.html
http://journals.witpress.com/pdfs/abstracts/D&NE040201a.pdf
http://blogs.christianpost.com/science-and-faith/pro-id-paper-examines-irreducible-complexity-of-birds-in-flight-9738/
http://creation.com/the-evolution-of-feathers-a-major-problem-for-darwinism
http://www.creationists.org/refuting-evolution-book-chapter-4.html
“THE IMPLICATION, THE RESEARCHERS SAID, IS THAT BIRDS ALMOST CERTAINLY DID NOT DESCEND FROM THEROPOD DINOSAURS, SUCH AS TYRANNOSAURUS OR ALLOSAURUS. THE FINDINGS ADD TO A GROWING BODY OF EVIDENCE IN THE PAST TWO DECADES THAT CHALLENGE SOME OF THE MOST WIDELY-HELD BELIEFS ABOUT ANIMAL EVOLUTION.
‘FOR ONE THING, BIRDS ARE FOUND EARLIER IN THE FOSSIL RECORD THAN THE DINOSAURS THEY ARE SUPPOSED TO HAVE DESCENDED FROM,’ RUBEN SAID.”
Mutation predicts 40 million years of fly wing evolution
This is remarkable when a paper in Nature magazine openly throws the towel and admits :

Our finding of striking similarities among mutational, genetic and
among-species variation coupled with a low rate of evolution are not readily explained by any of the available models of evolution. This is an important challenge for evolutionary theorists.
What is remarkable as well, is the misleading title of the paper....
http://www.nature.com.sci-hub.bz/nature/journal/vaop/ncurrent/full/nature23473.html?foxtrotcallback=true
The Genius of Birds
http://flightthegeniusofbirds.com/
Flight: The Genius of Birds - Official Trailer
https://www.youtube.com/watch?v=46rE4BfWN_M
Here's the bottom line we look at the anatomy of a bird its behavior its metabolism the structure of its feathers the structure of its muscles and so forth these are multiple independent points in a complex space out of which flight emerges and I think from a biological standpoint to fly at all requires a cause that's able to visualize a distant functional end point and bring together everything necessary to achieve that end point uniquely and universally in our experience only intelligence is capable of that kind of causal process an engineered system is the product of a mind that anticipated a problem and figured out a multi-step way of addressing that problem in birds you see exactly that kind of process they're engineering marvels they're works of art we know we're engineered things come from we know we're works of art come from so why would we attribute a bird to anything other than intelligence or mind
Last edited by Otangelo on Sun Oct 31, 2021 4:17 am; edited 20 times in total