Quantum entanglement holds and repairs together life’s blueprint
https://reasonandscience.catsboard.com/t2659-quantum-entanglement-holds-and-repairs-together-lifes-blueprint
Quantum entanglement holds together life’s blueprint
September 15, 2010
Entanglement is a quantum property where two or more objects are linked and in “superposition” – existing in many possible states at once. When this happens, it is impossible to describe the state of each object individually – the entangled objects must be considered as a whole. When the researchers analyzed the DNA without its helical structure, they found that the electron clouds were not entangled. But when they incorporated DNA’s helical structure into the model, they saw that the electron clouds of each base pair became entangled with those of its neighbors. “If you didn’t have entanglement, then DNA would have a simple flat structure, and you would never get the twist that seems to be important to the functioning of DNA,” says team member Vlatko Vedral of the University of Oxford. 2
The relevance of continuous variable entanglement in DNA
23 Feb 2011
We consider a chain of harmonic oscillators with dipole-dipole interaction between nearest neighbors resulting in a van der Waals type bonding. The binding energies between entangled and classically correlated states are compared. We apply our model to DNA. By comparing our model with numerical simulations we conclude that entanglement may play a crucial role in explaining the stability of the DNA double helix. 1
A base pair (bp) is a unit consisting of two nucleobases bound to each other by hydrogen bonds. 3 The base pairs GC and AT use hydrogen bonds rather than molecular bonds, or covalent bonds. Since DNA only uses hydrogen bonds, far less energy is required to unzip the chain. There’s far less likely that the DNA will get “cooked” in the process of unzipping or re-zipping DNA. Since these operations are done thousands of times a day, it’s very important that they work reliably with minimal chance of damaging the molecule. Hydrogen bonding happens in molecules that are dipoles: one end of the molecule has a positive charge and the other end has a negative charge. Water is one of the simplest molecules that work this way. Hydrogen bonding impacts all sorts of things from the phase of water (molecules this light are usually a gas at earth temperatures) to water’s surface tension to how ice forms on lakes. All of the behaviors are critical to life.
DNA and quantum entanglement 6
Jul 16, 2010
Does quantum entanglement hold DNA together? Some physicists say it’s possible.
Rieper and co ask what happens to these oscillations, or phonons as physicists call them, when the base pairs are stacked in a double helix.
Phonons are quantum objects, meaning they can exist in a superposition of states and become entangled, just like other quantum objects.
To start with, Rieper and co imagine the helix without any effect from outside heat. “Clearly the chain of coupled harmonic oscillators is entangled at zero temperature,” they say. They then go on to show that the entanglement can also exist at room temperature.
That’s possible because phonons have a wavelength which is similar in size to a DNA helix and this allows standing waves to form, a phenomenon known as phonon trapping. When this happens, the phonons cannot easily escape. A similar kind of phonon trapping is known to cause problems in silicon structures of the same size.
Quantum Entanglement stabilizes the DNA Molecular Structure 7
February 21, 2012
Van der Waals force
In physical chemistry, the van der Waals force (or van der Waals interaction), named after Dutch scientist Johannes Diderik van der Waals, is the sum of the attractive or repulsive forces between molecules (or between parts of the same molecule) other than those due to covalent bonds or to the electrostatic interaction of ions with one another or with neutral molecules. The term includes:
– force between two permanent dipoles (Keesom force)
– force between a permanent dipole and a corresponding induced dipole (Debye force)
– force between two instantaneously induced dipoles (London dispersion force)
It is also sometimes used loosely as a synonym for the totality of intermolecular forces. Van der Waals forces are relatively weak compared to normal chemical bonds. Van der Waals forces define the chemical character of many organic compounds. They also define the solubility of organic substances in polar and non-polar media. In low molecular weight alcohols, the properties of the polar hydroxyl group dominate the weak intermolecular forces of van der Waals. In higher molecular weight alcohols, the properties of the nonpolar hydrocarbon chain(s) dominate and define the solubility. Van der Waals-London forces grow with the length of the nonpolar part of the substance.

The Relevance Of Continuous Variable Entanglement In DNA
There was a time, not so long ago, when biologists thought that quantum mechanics could play no role in the hot, wet systems of life.
Since then, the discipline of quantum biology has emerged as one of the most exciting new fields in science. It’s beginning to look as if quantum effects are crucial in a number of biological processes, such as photosynthesis and avian navigation …
The new paradigm is called “Quantum Biology” and the evidence for quantum phenomenon occurring in “living things” at “room temperatures” is overwhelming. There is no way to deny how deep and profound the importance of quantum mechanics are in biological systems.
The structure of the DNA (helix) would vibrate and shake itself apart in a context where just pure classical laws of physics would reign. But all “base pairs” are held together by “phonons“, which exists in a superposition of states and become entangled, just like other quantum objects. So the result is that the vibration is zero because of the superposition state of all phonons in the DNA helix. They form a kind of “standing wave“.
How quantum entanglement in DNA synchronizes double-strand breakage by type II restriction endonucleases 5
2015 Dec 10
Macroscopic quantum effects in biological systems have been studied with verve in recent years, as researchers have sought fundamental explanations for diverse phenomena in bacteria, plants, flies, birds, and humans. Ensconced in thermally turbulent aqueous environments, biology appears to have found mechanisms to optimize structure and function for quantum behavior. My comment: " biology appears to have found": Hum, no. Biology is not sentient and does not find things. Teleology slips frequently into science commentaries - there is no justification for it. Orthodox type II restriction endonucleases cleave DNA in a manner that preserves the palindromic symmetry of the double-stranded substrates to which they bind. ( A palindromic sequence is a nucleic acid sequence on double-stranded DNA or RNA wherein reading 5' (five-prime) to 3' (three prime) forward on one strand matches the sequence reading 5' to 3' on the complementary strand with which it forms a double helix. ) Before cutting, these enzymes rapidly scan the DNA by facilitated diffusion searching for recognition sequences, which are between four and eight base pairs (bp) in length. Recognition sequence binding initiates conformational changes in the enzyme and DNA, releasing water and charge-countering ions from the protein-DNA interface. How sequence recognition proceeds to catalysis is perhaps the least understood aspect of the enzymology. Concerted cutting of both strands requires intersubunit correlations to synchronize the two catalytic centers. Under physiologically optimum conditions, several type II endonucleases demonstrate products that are cleaved entirely in both strands without producing intermediate single-strand cuts, suggesting a mechanism of synchronization between spatially separated nucleotides that is conserved in this class of enzymes. Such an absolute correlation over distance is a hallmark of quantum entanglement. Other quantum correlations—less prominent than entanglement but nonetheless without classical counterparts—have been quantified by the generalized concept of quantum discord and related measures, and these can indicate an advantage of quantum methods over classical ones even at higher temperatures. Type II restriction endonucleases that cut DNA in a concerted manner, as shown in Figure 1, would maintain quantum coherence in the DNA substrate by acting as a decoherence shield upon specific binding.

Proposed quantum entanglement in orthodox type II restriction endonuclease catalysis
(a) Enzyme (red) searches for recognition sequence on DNA (blue) by facilitated diffusion.
(b) Enzyme recognizes target site, undergoing a conformational change to tightly bind the DNA sequence and form the Decoherence-free subspaces . Clamping induces excitation of quantized oscillations from coupled base-pair electron clouds (green), entangling two electrons in phosphodiester bonds (orange) on opposing strands of the helix.
(c) Synchronized catalysis occurs as quanta decay symmetrically into the entangled bonds, thus breaking the DNA helix in a single binding event.
Decoherence shields have been evoked in the quantum biology literature. Conformational change induced between nonspecific and specific binding is commensurate with the exclusion of ions and over 100 water molecules from the surface of DNA. Squeezing water away from the DNA helix may be the procedure by which some type II endonucleases create decoherence-free subspaces for quantum entanglement to occur. Release of ions and charge cancellation by amino acid residues would minimize electromagnetic interaction with the delicately shielded quantum state.
biology the designer has dynamically optimized several parameters to achieve maintenance of the proposed DNA quantum state.
Quantum entanglement helps keep DNA together 4
JUNE 29, 2010
Once in a while science produces theoretical work that has tantalizing possibilities but also raises a strong skeptical response. This is another way of saying that a theory has a certain amount of plausibility but is without experimental evidence. Such is the case with a theory proposed by Elisabeth Rieper and colleagues at the National University of Singapore and submitted in a paper at arXiv.org on June 21, 2010: The relevance of continuous variable entanglement in DNA. They are saying that the stability of DNA is in part the result of quantum entanglement.
It’s a little early to be talking about the ‘field’ of quantum biology, but if the existence of significant quantum effects in DNA can be substantiated (that means with experimental evidence) it would be a foundational discovery. However, at this point the idea is a working hypothesis based on mathematical modeling. It goes something like this…
Quantum effects, in this case quantum entanglement, are among the most counter-intuitive and challenging ideas in physics – ‘spookish’ is the word Einstein used. Scientists know quantum mechanics from many decades of mathematical theorizing and a couple of decades of experimentation with atomic behavior at temperatures close to absolute zero. It comes as an enormous intellectual and theoretical leap to grapple with the idea that natural (biological) phenomenon use quantum effects at or above room temperature. Yet, the evidence is accumulating that quantum entanglement is involved with biology in a very fundamental way – photosynthesis being the first to have experimental evidence.
This new theory, which is certain to provoke as many skeptical voices as words of interest, began with wondering what role, if any, might quantum entanglement play in DNA. Quantum entanglement is described, simply, as two separate particles that work together as if they were one particle no matter how far apart they might be. If one particle moves up, the other particle moves down, instantly, as if they were on the ends of a teeter-totter. They are a system that behaves as one particle. In the case of DNA, the ‘particles’ are the molecules of the DNA base pairs, formed by the nucleotides with adenine, guanine, thymine, and cytosine. Each nucleotide is surrounded by a cloud of electrons that behave as if the nucleotide were an atomic nucleus. The cloud shifts relative to the nucleus, perhaps influenced by what are called Van der Waals forces, from side to side so to speak forming a dipole (two poles), and this shifting is regular – a harmonic oscillation. In solid-state physics, the oscillation of molecules within a solid is known as a phonon, a kind of quasi-particle that vibrates at a specific frequency and gives the solid many of its electrical and physical properties. In DNA, when a base pair is formed the clouds of each nucleotide must oscillate in opposite directions if the bond is to hold together.
The key question for the researchers was what influence does the double helical structure of DNA have on this oscillation? To answer the question, they first modeled how the phonons would behave at absolute zero temperature. Here (mathematically) it was clear the phonons would be typical quantum objects, existing as both waves and particles exhibiting the property of quantum entanglement. As it turns out, the size of the DNA helix corresponds rather well to the wavelength (frequency) of the phonons. This correspondence causes the phonons to stay within this frequency, something called ‘phonon trapping.’ Though the nucleotide phonons in each base pair oscillate in opposite directions they do so in a quantum entangled system – they act together and at the same frequency, ensuring the stability of the pair bond and of the helix itself.
At least that’s what the model shows can happen. The model also shows that this configuration can maintain the bond at high temperatures – room temperatures or above (e.g. 20 degrees C or 68 degrees F). The quantum entanglement is vital to making this work, because under classical mechanics the vibration of the particles in the helix would shake it apart, especially at higher temperatures.
Of course, this is all modeling. What must come next is experimental evidence. It won’t come easily. The researchers point to the notion that using classical mechanics to add up the energy necessary to hold the helix together comes out short, and that adding the quantum effects makes up the difference. But this is indirect evidence.
Keep in mind that quantum mechanics existed as mathematical theory long before experimental evidence was provided. The situation may be similar with showing quantum effects in DNA. Or not. As is the case when there are potentially major shifts in scientific understanding – and finding quantum mechanics as a basis for some of the most fundamental aspects of biology certainly qualifies – the demands for evidence will be rigorous. In the meantime, scientists will engage in vigorous debate. It will be interesting to see how argument and evidence changes or nullifies the theoretical insight. The process represents the essence of the scientific method applied to a potentially revolutionary
1. https://arxiv.org/abs/1006.4053v1
2. https://neshealthblog.wordpress.com/2010/09/15/quantum-entanglement-holds-together-lifes-blueprint/
3. https://en.wikipedia.org/wiki/Base_pair
4. https://web.archive.org/web/20160331105908/http://scitechstory.com/2010/06/29/quantum-entanglement-helps-keep-dna-together/
5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4746125/
6. https://kottke.org/10/07/dna-and-quantum-entanglement
7. https://www.hydrogen2oxygen.net/en/quantum-entanglement-stabilizes-the-dna-molecular-structure/
https://reasonandscience.catsboard.com/t2659-quantum-entanglement-holds-and-repairs-together-lifes-blueprint
Quantum entanglement holds together life’s blueprint
September 15, 2010
Entanglement is a quantum property where two or more objects are linked and in “superposition” – existing in many possible states at once. When this happens, it is impossible to describe the state of each object individually – the entangled objects must be considered as a whole. When the researchers analyzed the DNA without its helical structure, they found that the electron clouds were not entangled. But when they incorporated DNA’s helical structure into the model, they saw that the electron clouds of each base pair became entangled with those of its neighbors. “If you didn’t have entanglement, then DNA would have a simple flat structure, and you would never get the twist that seems to be important to the functioning of DNA,” says team member Vlatko Vedral of the University of Oxford. 2
The relevance of continuous variable entanglement in DNA
23 Feb 2011
We consider a chain of harmonic oscillators with dipole-dipole interaction between nearest neighbors resulting in a van der Waals type bonding. The binding energies between entangled and classically correlated states are compared. We apply our model to DNA. By comparing our model with numerical simulations we conclude that entanglement may play a crucial role in explaining the stability of the DNA double helix. 1
A base pair (bp) is a unit consisting of two nucleobases bound to each other by hydrogen bonds. 3 The base pairs GC and AT use hydrogen bonds rather than molecular bonds, or covalent bonds. Since DNA only uses hydrogen bonds, far less energy is required to unzip the chain. There’s far less likely that the DNA will get “cooked” in the process of unzipping or re-zipping DNA. Since these operations are done thousands of times a day, it’s very important that they work reliably with minimal chance of damaging the molecule. Hydrogen bonding happens in molecules that are dipoles: one end of the molecule has a positive charge and the other end has a negative charge. Water is one of the simplest molecules that work this way. Hydrogen bonding impacts all sorts of things from the phase of water (molecules this light are usually a gas at earth temperatures) to water’s surface tension to how ice forms on lakes. All of the behaviors are critical to life.
DNA and quantum entanglement 6
Jul 16, 2010
Does quantum entanglement hold DNA together? Some physicists say it’s possible.
Rieper and co ask what happens to these oscillations, or phonons as physicists call them, when the base pairs are stacked in a double helix.
Phonons are quantum objects, meaning they can exist in a superposition of states and become entangled, just like other quantum objects.
To start with, Rieper and co imagine the helix without any effect from outside heat. “Clearly the chain of coupled harmonic oscillators is entangled at zero temperature,” they say. They then go on to show that the entanglement can also exist at room temperature.
That’s possible because phonons have a wavelength which is similar in size to a DNA helix and this allows standing waves to form, a phenomenon known as phonon trapping. When this happens, the phonons cannot easily escape. A similar kind of phonon trapping is known to cause problems in silicon structures of the same size.
Quantum Entanglement stabilizes the DNA Molecular Structure 7
February 21, 2012
Van der Waals force
In physical chemistry, the van der Waals force (or van der Waals interaction), named after Dutch scientist Johannes Diderik van der Waals, is the sum of the attractive or repulsive forces between molecules (or between parts of the same molecule) other than those due to covalent bonds or to the electrostatic interaction of ions with one another or with neutral molecules. The term includes:
– force between two permanent dipoles (Keesom force)
– force between a permanent dipole and a corresponding induced dipole (Debye force)
– force between two instantaneously induced dipoles (London dispersion force)
It is also sometimes used loosely as a synonym for the totality of intermolecular forces. Van der Waals forces are relatively weak compared to normal chemical bonds. Van der Waals forces define the chemical character of many organic compounds. They also define the solubility of organic substances in polar and non-polar media. In low molecular weight alcohols, the properties of the polar hydroxyl group dominate the weak intermolecular forces of van der Waals. In higher molecular weight alcohols, the properties of the nonpolar hydrocarbon chain(s) dominate and define the solubility. Van der Waals-London forces grow with the length of the nonpolar part of the substance.

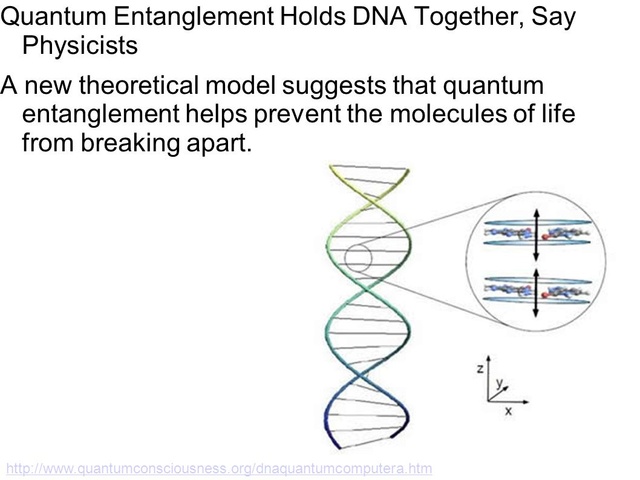
The structure of the DNA double helix. The atoms in the structure are colour coded by element and the detailed structure of two base pairs is shown in the bottom right.
The Relevance Of Continuous Variable Entanglement In DNA
There was a time, not so long ago, when biologists thought that quantum mechanics could play no role in the hot, wet systems of life.
Since then, the discipline of quantum biology has emerged as one of the most exciting new fields in science. It’s beginning to look as if quantum effects are crucial in a number of biological processes, such as photosynthesis and avian navigation …
The new paradigm is called “Quantum Biology” and the evidence for quantum phenomenon occurring in “living things” at “room temperatures” is overwhelming. There is no way to deny how deep and profound the importance of quantum mechanics are in biological systems.
The structure of the DNA (helix) would vibrate and shake itself apart in a context where just pure classical laws of physics would reign. But all “base pairs” are held together by “phonons“, which exists in a superposition of states and become entangled, just like other quantum objects. So the result is that the vibration is zero because of the superposition state of all phonons in the DNA helix. They form a kind of “standing wave“.
How quantum entanglement in DNA synchronizes double-strand breakage by type II restriction endonucleases 5
2015 Dec 10
Macroscopic quantum effects in biological systems have been studied with verve in recent years, as researchers have sought fundamental explanations for diverse phenomena in bacteria, plants, flies, birds, and humans. Ensconced in thermally turbulent aqueous environments, biology appears to have found mechanisms to optimize structure and function for quantum behavior. My comment: " biology appears to have found": Hum, no. Biology is not sentient and does not find things. Teleology slips frequently into science commentaries - there is no justification for it. Orthodox type II restriction endonucleases cleave DNA in a manner that preserves the palindromic symmetry of the double-stranded substrates to which they bind. ( A palindromic sequence is a nucleic acid sequence on double-stranded DNA or RNA wherein reading 5' (five-prime) to 3' (three prime) forward on one strand matches the sequence reading 5' to 3' on the complementary strand with which it forms a double helix. ) Before cutting, these enzymes rapidly scan the DNA by facilitated diffusion searching for recognition sequences, which are between four and eight base pairs (bp) in length. Recognition sequence binding initiates conformational changes in the enzyme and DNA, releasing water and charge-countering ions from the protein-DNA interface. How sequence recognition proceeds to catalysis is perhaps the least understood aspect of the enzymology. Concerted cutting of both strands requires intersubunit correlations to synchronize the two catalytic centers. Under physiologically optimum conditions, several type II endonucleases demonstrate products that are cleaved entirely in both strands without producing intermediate single-strand cuts, suggesting a mechanism of synchronization between spatially separated nucleotides that is conserved in this class of enzymes. Such an absolute correlation over distance is a hallmark of quantum entanglement. Other quantum correlations—less prominent than entanglement but nonetheless without classical counterparts—have been quantified by the generalized concept of quantum discord and related measures, and these can indicate an advantage of quantum methods over classical ones even at higher temperatures. Type II restriction endonucleases that cut DNA in a concerted manner, as shown in Figure 1, would maintain quantum coherence in the DNA substrate by acting as a decoherence shield upon specific binding.

Proposed quantum entanglement in orthodox type II restriction endonuclease catalysis
(a) Enzyme (red) searches for recognition sequence on DNA (blue) by facilitated diffusion.
(b) Enzyme recognizes target site, undergoing a conformational change to tightly bind the DNA sequence and form the Decoherence-free subspaces . Clamping induces excitation of quantized oscillations from coupled base-pair electron clouds (green), entangling two electrons in phosphodiester bonds (orange) on opposing strands of the helix.
(c) Synchronized catalysis occurs as quanta decay symmetrically into the entangled bonds, thus breaking the DNA helix in a single binding event.
Decoherence shields have been evoked in the quantum biology literature. Conformational change induced between nonspecific and specific binding is commensurate with the exclusion of ions and over 100 water molecules from the surface of DNA. Squeezing water away from the DNA helix may be the procedure by which some type II endonucleases create decoherence-free subspaces for quantum entanglement to occur. Release of ions and charge cancellation by amino acid residues would minimize electromagnetic interaction with the delicately shielded quantum state.
CONCLUSIONS
Just as complex systems exhibit behavior that cannot be predicted from the mechanics of microscopic constituents, soQuantum entanglement helps keep DNA together 4
JUNE 29, 2010
Once in a while science produces theoretical work that has tantalizing possibilities but also raises a strong skeptical response. This is another way of saying that a theory has a certain amount of plausibility but is without experimental evidence. Such is the case with a theory proposed by Elisabeth Rieper and colleagues at the National University of Singapore and submitted in a paper at arXiv.org on June 21, 2010: The relevance of continuous variable entanglement in DNA. They are saying that the stability of DNA is in part the result of quantum entanglement.
It’s a little early to be talking about the ‘field’ of quantum biology, but if the existence of significant quantum effects in DNA can be substantiated (that means with experimental evidence) it would be a foundational discovery. However, at this point the idea is a working hypothesis based on mathematical modeling. It goes something like this…
Quantum effects, in this case quantum entanglement, are among the most counter-intuitive and challenging ideas in physics – ‘spookish’ is the word Einstein used. Scientists know quantum mechanics from many decades of mathematical theorizing and a couple of decades of experimentation with atomic behavior at temperatures close to absolute zero. It comes as an enormous intellectual and theoretical leap to grapple with the idea that natural (biological) phenomenon use quantum effects at or above room temperature. Yet, the evidence is accumulating that quantum entanglement is involved with biology in a very fundamental way – photosynthesis being the first to have experimental evidence.
This new theory, which is certain to provoke as many skeptical voices as words of interest, began with wondering what role, if any, might quantum entanglement play in DNA. Quantum entanglement is described, simply, as two separate particles that work together as if they were one particle no matter how far apart they might be. If one particle moves up, the other particle moves down, instantly, as if they were on the ends of a teeter-totter. They are a system that behaves as one particle. In the case of DNA, the ‘particles’ are the molecules of the DNA base pairs, formed by the nucleotides with adenine, guanine, thymine, and cytosine. Each nucleotide is surrounded by a cloud of electrons that behave as if the nucleotide were an atomic nucleus. The cloud shifts relative to the nucleus, perhaps influenced by what are called Van der Waals forces, from side to side so to speak forming a dipole (two poles), and this shifting is regular – a harmonic oscillation. In solid-state physics, the oscillation of molecules within a solid is known as a phonon, a kind of quasi-particle that vibrates at a specific frequency and gives the solid many of its electrical and physical properties. In DNA, when a base pair is formed the clouds of each nucleotide must oscillate in opposite directions if the bond is to hold together.
The key question for the researchers was what influence does the double helical structure of DNA have on this oscillation? To answer the question, they first modeled how the phonons would behave at absolute zero temperature. Here (mathematically) it was clear the phonons would be typical quantum objects, existing as both waves and particles exhibiting the property of quantum entanglement. As it turns out, the size of the DNA helix corresponds rather well to the wavelength (frequency) of the phonons. This correspondence causes the phonons to stay within this frequency, something called ‘phonon trapping.’ Though the nucleotide phonons in each base pair oscillate in opposite directions they do so in a quantum entangled system – they act together and at the same frequency, ensuring the stability of the pair bond and of the helix itself.
At least that’s what the model shows can happen. The model also shows that this configuration can maintain the bond at high temperatures – room temperatures or above (e.g. 20 degrees C or 68 degrees F). The quantum entanglement is vital to making this work, because under classical mechanics the vibration of the particles in the helix would shake it apart, especially at higher temperatures.
Of course, this is all modeling. What must come next is experimental evidence. It won’t come easily. The researchers point to the notion that using classical mechanics to add up the energy necessary to hold the helix together comes out short, and that adding the quantum effects makes up the difference. But this is indirect evidence.
Keep in mind that quantum mechanics existed as mathematical theory long before experimental evidence was provided. The situation may be similar with showing quantum effects in DNA. Or not. As is the case when there are potentially major shifts in scientific understanding – and finding quantum mechanics as a basis for some of the most fundamental aspects of biology certainly qualifies – the demands for evidence will be rigorous. In the meantime, scientists will engage in vigorous debate. It will be interesting to see how argument and evidence changes or nullifies the theoretical insight. The process represents the essence of the scientific method applied to a potentially revolutionary

1. https://arxiv.org/abs/1006.4053v1
2. https://neshealthblog.wordpress.com/2010/09/15/quantum-entanglement-holds-together-lifes-blueprint/
3. https://en.wikipedia.org/wiki/Base_pair
4. https://web.archive.org/web/20160331105908/http://scitechstory.com/2010/06/29/quantum-entanglement-helps-keep-dna-together/
5. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4746125/
6. https://kottke.org/10/07/dna-and-quantum-entanglement
7. https://www.hydrogen2oxygen.net/en/quantum-entanglement-stabilizes-the-dna-molecular-structure/


